In this article, we have given complete list of nursing notes to prepare for nursing examination (BSC, GNM, P.C. (P.B) BSC AND MSC NURSING). The nursing notes prepared according to the NURSING SYLLABUS.
CONCURRENT & TERMINAL DISINFECTION
TERMINAL DISINFECTION Concurrent & Terminal Disinfection – Steps and Procedure
UPDATED 2024
Terminal disinfection typically refers to the thorough disinfection of surfaces and equipment in a healthcare setting, particularly in areas such as patient rooms, operating rooms, and other high-risk areas where the spread of infections can occur. The goal is to eliminate or reduce the presence of pathogens, including bacteria and viruses, to prevent the transmission of infections.
Here are some general steps and considerations for terminal disinfection in healthcare settings:
- Personal Protective Equipment (PPE): Ensure that individuals performing the disinfection process wear appropriate PPE, including gloves and, if necessary, masks and gowns.
- Cleaning: Before disinfection, surfaces should be cleaned to remove any visible dirt or organic material. Use a detergent or cleaning solution appropriate for the surfaces being cleaned.
- Disinfection Agents: Choose an appropriate disinfectant based on the type of pathogens you are targeting. Common disinfectants include quaternary ammonium compounds, hydrogen peroxide, and bleach. Follow the manufacturer’s instructions for dilution and contact time.
- Application Method: Apply the disinfectant according to the recommended method. This could involve using wipes, sprays, or other application devices. Ensure thorough coverage of all surfaces, especially high-touch areas.
- Contact Time: Allow the disinfectant to remain on surfaces for the recommended contact time. This is the time the disinfectant needs to be in contact with the surface to effectively eliminate pathogens.
- Ventilation: Ensure proper ventilation in the area being disinfected. This helps to reduce the concentration of disinfectant fumes and aids in the drying of surfaces.
- Equipment and Furniture: Disinfect all movable equipment and furniture in the room, including bedrails, tables, and medical devices. Pay extra attention to high-touch surfaces.
- Waste Disposal: Dispose of disposable items, cleaning materials, and any waste generated during the disinfection process according to established protocols.
- Post-Disinfection Inspection: After the disinfection process, perform an inspection to ensure that all surfaces are adequately disinfected. Re-clean and re-disinfect any areas that may have been missed.
- Documentation: Maintain records of the disinfection process, including the disinfectant used, concentration, contact time, and any issues encountered during the process.
DISINFECTION OF ARTICLES
Disinfection means destroying of all the pathogenic organisms. It is done either by physical or chemical method.
Types of Disinfectants
- Alcohols
- Ethanol
- Isopropanol
Advantages: wide microbicidal activity, noncorrosive
Disadvantages: not universally sporicidal, limited residual activity, fire hazard
Recommended for: hand disinfection in outpatient clinics
- Aldehyde
- Glutaraldehyde
- Orthophthaldehyde
Advantages: wide microbicidal activity and are sporicidal and fungicidal
Disadvantages: glutaraldehyde is potentially a toxic health hazard
Recommended for: 2% glutaraldehyde with alkaline pH used for disinfecting laminated non-autoclavable equipments
Orthphthaldehyde replacing glutaraldehyde as a nontoxic disinfectant
- Oxidizing agents
- Sodium hypochlorite
Advantages: wide microbicidal activity and are sporicidal and fungicidal
Disadvantages: corrosive
Recommended for: useful for disinfecting surfaces, water contamined equipments
- Hydrogen peroxide
Advantages: wide microbicidal activity and are sporicidal and fungicidal
Disadvantage: unstable compound; to be used in correct concentration
Recommended for: used for logging – high level disinfection of Operation Theater when used in concentration of over 6%
- Phenolics
Advantage: easily available, cheap
Disadvantage: corrosive to the skin and sometimes toxic to sensitive people. Household disinfectants
Recommended for: resistant organisms are common
Suitable for surface disinfection of residential premises
- Quaternary ammonium compounds
Advantages: active against enveloped viruses
Disadvantages: low sporicidal activity
Recommended for: low level disinfectants
Disinfectant
- Should be efficient
- Should be used in the correct strength
- Should be applied for a sufficient length of time
- Should not be injurious to the articles
- The article should be fully immersed in it
CONCURRENT DISINFECTION
Concurrent disinfection is the immediate disinfection of all contaminated articles and bodily discharges during the course of the disease
Concurrent disinfection of the contaminated articles immediately, then and there during the course of illness. It includes cleaning the isolation unit daily with effective disinfectant
- Urinals and bedpans: after emptying clean with brush soak in carbolic solution 1:40 for disinfection of articles: 10 minutes
- Sputum mugs: collect the sputum in a sputum mug with disinfectant lotion. When container is full, it should be boiled or buried
- Linen: keep the linen in a carbolic solution or any other effective disinfectant for 4 hours rinse, dry and send to laundry
- Blankets: autoclaving is the best
- Mattresses and pillow: place in direct sunlight for 6 hours for two consecutive days
- Mackintosh: soap in Lysol or phenol 1:40 or 4 hours. Wash and dry in shade
- Paper, cotton swabs, books: burn in disposal of all wastes by incineration
- Disposal of excreta: especially for enteric isolation the urine and stool should be mixed with equal quantity of ( 1 part of lime to 4 parts of water) and allowed to stand for two to four hours. Then disposed by burial
Disinfectant should not be added in stool and discarded in septic tank as the disinfectant hinders the natural biological action
TERMINAL DISINFECTION
Terminal disinfection means disinfection of the patients unit and all the articles used after the discharge, transfer or death of the patient who was suffering from the contagious disease
It means after the patient has recovered from a communicable disease, the disinfection of the room and articles used by him is done after his discharge from the unit or transfer or death
Fumigation with sulfur: fumigation is disinfection by exposure to the fumes of a vaporized disinfectant or use of fumes or gases to destroy organisms. Fumigation with sulfur- the room should be filled with steam form a kettle. Sulfur in an earthen ware which is kept in a large container with water. Pour methylated spirit over the sulphur, put the sulphur afire and close the room
Fumigation with formalin: it is done with potassium permanganate crystals and formalin is used for this purpose. Mix and place them in a metal bowl. For 100 cu feet, 140 gm of KMO4 and 250 ml of formalin are to be used then room should be closed completely and seated for 24 hours for effective disinfection
Isolation technique, a medical aseptic practice, inhibits the spread and transfer of pathogenic organisms by limiting the contacts of the patient and creating some kind of physical barrier between the patient and others. In isolation techniques, disinfection procedures are employed to control contaminated items and areas.
Disinfection is described as the killing of certain infectious (pathogenic) agents outside the body by a physical or chemical means. Isolation techniques employ two kinds of disinfection practices, concurrent and terminal

CARDIOPULMONARY RESUSCITATION (CPR)
CARDIOPULMONARY RESUSCITATION (CPR) (Definition, Purpose, Equipment, General Instructions, Procedure, Method, Do’s and don’ts in CPR and Complications.
UPDATED 2024
Cardiopulmonary resuscitation is a lifesaving technique useful in many emergencies, including heart attack or near drowning, in which someone’s breathing or heartbeat has stopped. The American Heart Association recommends that everyone – untrained bystanders and medical personnel alike – begin CPR with chest compressions.
Resuscitation includes all measures that are applied to revive patients who have stopped breathing suddenly and unexpectedly due to either respiratory or cardiac failure.
Cardiac arrest is one of the common causes for cardio-respiratory failure. When a person stops breathing spontaneously, his heart also stops beating. Clinical death occurs within 4-6 minutes, the cells of the brain which is sensitivity to the paucity of oxygen begin to deteriorate. If the oxygen supply is not restored, the patient suffers irreversible brain damage and biological death occurs.
DEFINTION
Artificial ventilation accompanied by cardiac massage to facilitate normal breathing and heart action in the event of cardiac arrest.
PURPOSE
To re-establish effective ventilation and circulation
EQUIPMENT
- Cardiac board
- Suction apparatus
- Oxygen supply
- Box containing Ambu bag
- Sterile endotracheal tube (2.5 – 5.5 mm)
- Extra-batteries
- Laryngoscope with 0, 1, 2 size tongue blades and stillet, Magill forceps, adhesive scissors, airway syringes 1, 2, 5, 10 cc
- Intracardiac needle 20 G, 22 G, 6-8 cm length
- Needles 23 G and 20 G
- Elastoplasts bandage
- Ventilation given with ambu-bag
GENERAL INSTRUCTIONS
- Identify “RED FLAG” signs of critically ill child-changes in level of consciousness, flaccid posturing, cyanosis severe chest retractions, grunting respiration, increased respiratory rate, shallow respiration, see saw respiration, i.e. abdominal protrusion with inhalation, irregular respirations with periodic deep sighs, apneas, absent pulse, absent heart rate, absent carotid pulse, dilated pupils, unrecordable blood pressure, cold clammy skin
- ACT quickly! As child can go into cerebral hypoxia within 3 to 4 minutes which will lead to permanent brain damage
- Assess child (look, listen, feel) and if not breathing call for help
- Immediately start cardiopulmonary resuscitation (CPR)
- Equipment for CPR to be always accessible and is functioning condition
- All CPR equipment to be checked at beginning of each shift
- All staff to be skillful at CPR
PROCEDURE
- Airway: establish patient airway by suctioning oropharynx with catheter, and deflate stomach by aspirating stomach contents
Ventilation by mouth to mouth:
- Breathing: establish breathing by artificial ventilation
Place
Ambu bag on mouth and nose, and connect to 100% oxygen. Select ET tube using the formula:
Age in years + 4 /4
Calculate size of ET tube approximately as diameter of child’s little finger. The ET tube is inserted
- Circulation: initiate cardiac compression to a distance calculated using the formula (ET size multiply 3 cm)
METHOD
Serial rhythmic compressions of chest that help circulate oxygen containing blood to vital organs
Infant
- Site: sternum compression – below level of infant’s nipples
- Width one finger breadth
- Depth 0.5 – 1 inch
- Rate 100 times per minute
Child
- Site: lower margin of child’s rib cage to notch where ribs and sternum meet
- Avoid compression over notch
- Place heel of nurse’s hand over lower half of sternum (between nipple line and notch)
- Depth: 1-1.5 inches
- Rate: 100 times per minute
Ratio of Cardiac Compression to Ventilation (CPR)
- 2 persons – 5:1
- 1 person – 15:2
One Rescuer CPR
Shake shoulders and ask “are you okay”, shout for help. Open the airway: the most important action for successful resuscitation is immediate opening of the airway. Tilt the head by applying firm backward pressure on the victim’s forehead with palm of one hand. Place two or three fingers of the other hand under the bony part of the lower jaw near the chin and lift the chin
Check for breathing: please check close to victim’s mouth and nose. Look at chest to see if it rises and falls. Listen and feel for exhaled air (for at least 5 seconds)
External Cardiac Massage
Breathe: maintain an open airway. Pinch nose. Seal lips around victims mouth and deliver two full breathes watching chest to rise and fall with each breath
Check for circulation: feel for a carotid pulse. Again shout for help/activate EMS system. If pulse is present, continue to give artificial ventilation at the rate of 1 breath or 12 mm
Circulate: if pulse is absent, run fingers along the lower rib to notch in centre of the heart where ribs meet sternum. With middle finger in notch, place index finger on lower end of sternum. Place heel of other hand on lower ½ of sternum next to index finger. Put the heel of 1st hand on top. With shoulders directly over sternum and elbows locked, compress straight up and down 15 minutes, at the rate of 80 – 100 times a minute, using the count “one and two and three and”, etc. return quickly to victims head to deliver two breaths. Compression depth should be 1.5 – 2 inches
Two-rescuer CPR: two medical professional arriving at same time – no
CPR in Progress
- First rescuer
Determine unresponsiveness
Opens the airway
Checks for breathing
Ventilates twice, watching chest movement
Checks for carotid pulse: give command to begin compressions if pulse is absent
- Second rescuer
Locates landmark and proper hand position on sternum
Begins chest compressions on command – at rate of 80-100 per minute, counting “one and two and three and four and five and”
Pauses after each fifth compression to allow for ventilation
Calls for a switch when fatigued. Give clear signal “change and two and three and four and five”
- Both rescuers change simultaneously
Compression moves to victim’s head. After checking for pulse, give breath and command to continue compressions
Ventilator moves to chest: finds landmark and properly positions hands, begins compressions on command pausing after each 5th compression for breath
If CPR is in progress by lay person, rescue team enters after completion of cycle of 15 compressions and 2 ventilations and start with a reassessment
If CPR is in progress by a professional rescuer, the 2nd professional rescuer takes over compressions at the end of a cycle and after 1st rescuer reassesses pulse and gives another breath
DO’S AND DONOT’S IN CPR
DO’S
- Reassure victim that help is on the way
- For major injuries call 9-1-1 immediately
- Check victim’s status regularly
- Use direct pressure to stop bleeding
- Check to see if victim’s airways are clear
- If no pulse or respiration, start CPR
- To prevent transmission of disease, use latex gloves
- Keep victims in shock warm (use blanket, etc)
- Assume spinal injury when blunt force trauma is present
- Raise head if bleeding in upper torso area
- Raise feet if bleeding in lower torso areas
- Flush all burns and chemical injuries with clean water
- Have MSDS sheets on the jobsite for 9-1-1 responders
- Call the Poison Control Center for chemical ingestion
DON’TS
- Do not move the victim unless absolutely necessary
- Always suspect “spinal injury” (and don’t move the victim)
- Do not set fractures and breaks (simply immobilize the victim)
- Do not apply a tourniquet (use “direct” pressure to stop bleeding)
- Do not remove items imbedded in the eye (cover with a Dixie cup)
- Do not use burn ointments
- Do not hesitate to call 9-1-1
COMPLICATIONS
Cardiopulmonary resuscitation, or CPR, is a technique used to support the circulation of blood and oxygen in the body of a victim who is not breathing and does not have a pulse. CPR is physically invasive for the victim and techniques used during CPR carry risks and the chance of complications. Ultimately, the risk of complications is small and should not deter the use of CPR for a victim in need
Broken bones
Rib fractures are the most common complication of CPR. Chest compressions administered during CPR are given quickly and with enough force to compress the chest about 1 inch in depth. This provides pressure to the ribs, which can be strong enough to cause ribs to fracture. Victims who are elderly, small in stature or children have the highest risk of developing rib fracture during chest compressions. Additionally, the chest bone, or sternum, also endures pressure and stress during chest compressions and can fracture as well
Internal Injuries
Internal organs lie within the area pressured by chest compressions. As the chest is compressed during CPR, ribs and chest bones can break, puncturing the lungs and liver. Additionally, internal bruising of the heart and liver can occur
Vomiting and Aspiration
As chest compressions are administered, pressure builds inside the body, which can force stomach contents up the esophagus and result in vomiting. This causes the risk of aspiration, or absorbing the vomit into the respiratory system. Aspiration is a serious complication which makes it difficult to provide the victim with adequate air and can ultimately damage lung tissue or result in infection, like pneumonia.
Body Fluid Exposure
CPR presents the risk of exposure to body fluids. It provides mouth-to-mouth rescue breathing to a victim without use of a mask results in saliva exposure between victim and rescuer. Blood and vomit may also be present during CPR, which carries the risk of communicable disease such as hepatitis and AIDS. The American Heart Association encourages the use of a barrier mask when administering rescue breathing during CPR for protection against contamination
Gastric Distention
Rescue breathing during CPR provides air directly into the lungs of the victim. If air is delivered too forcefully or for too long a time, the victim can accumulate air build-up in the stomach, called gastric distension. Gastric distension causes the stomach to swell and places pressure on the lungs. CPR efforts can become complicated if gastric distension occurs due to reduced ability to deliver adequate oxygen to the lungs, and can also result in vomiting and aspiration. Gastric distension can often be avoided by proper, careful administration of rescue breathing during CPR

BACK CARE / BACK MASSAGE / BACK RUB
BACK CARE/BACK MASSAGE/BACK RUB
UPDATED 2024
Back care means cleaning and massaging back, paying special attention to pressure points
Back massage provides comfort pleases and relaxes the patient; thereby it facilitates the physical stimulation to the skin and the emotional relaxation
Back rub means attending the back and pressure points of body with special care it is often called as back care or back massage
Purpose
- To give comfort to the patient
- To stimulate blood circulation
- To promote rest and sleep
- To prevent pressure sores
- To assess the skin condition
- To relax and relieve tension in tissues and muscles
- To refresh patient and relieve fatigue
General Instructions
- Back care given as a part of morning care and evening care
- Pressure points are attended more frequently and the position is changed
- When the skin is greasy, moist, thin about to break or patient is in continent or edematous used spirit or powder to reduce friction
- When the skin is dry, use oil for back rub. Spirit toughens the skins and powder reduces friction oil lubricates the skin and, hence friction
- When giving back rub, use more pressure on upward strokes towards the head and less pressure on the downward strokes
- Back rub may be contraindicated in patients susceptible to clotting disorders
Equipment
A tray containing of:
- A basin of warm water
- Sponge cloths – 2
- Soap and towel
- Surgical spirit or back-rub lotion and powder
- Mackintosh and towel
- Kidney tray and paper bag
Procedure
- Wash hands and explain the procedure
- Screen the patient and explain the procedure
- Turn the patient on his side
- Turn back top bedding and expose only required part
- Spread towel close to the patients back to protect bed linen
- Wash back thoroughly from cervical spine to the coccyx
- Apply soap in the same manner. Run hands firmly and slowly up the back on either side of the vertebral column up the neck and down across the shoulders
- Pour some spirit in to hand applies firmly in a circular motion repeat until back is thoroughly rubbed with it
- Wash off soap and dry thoroughly with towel
- The back must be rubbed three to five minutes especially over pressure points
- Apply back powder after through drying of the spirit
- Remove the towel
- Cover the patient with top bedding
After Care
- Make the patient comfortable
- Remove the screen and equipment
- Clean the articles with soap and water and keep ready for next use
- Wash hands
- Record the date, time treatment and observation made on nurse’s record

DIABETES MELLITUS
DIABETES MELLITUS – General Characteristics, Pancreas, Classification, Etiopathogenesis, Pathological Changes, Clinical Features, Diagnosis and Treatment
UPDATED 2024
General Characteristics
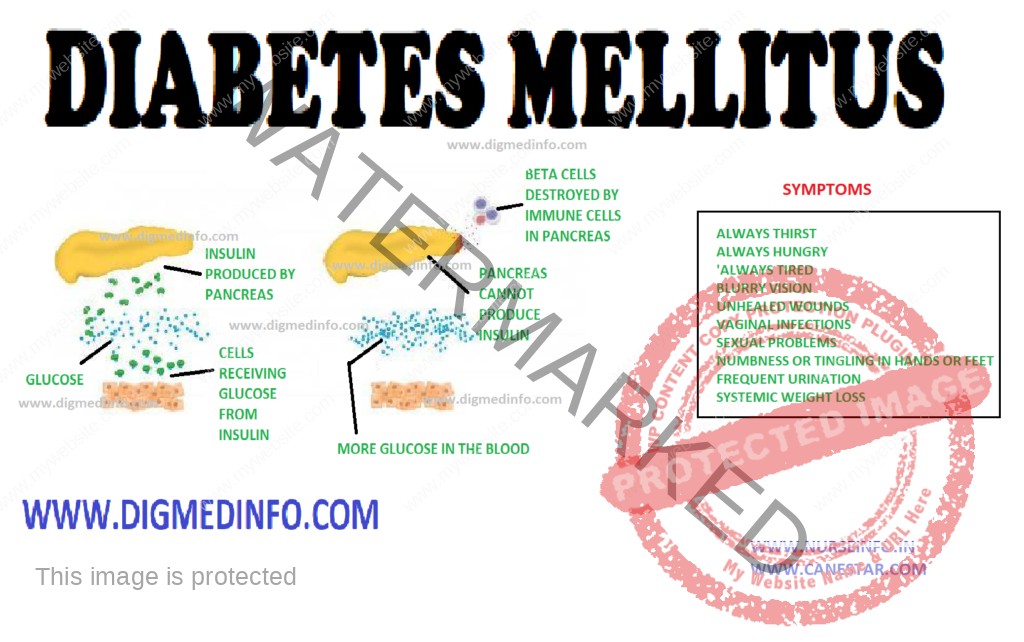
Diabetes mellitus is a chronic metabolic disorder characterized by hyperglycemia with or without glycosuria, resulting from an absolute or relative deficiency of insulin. This is brought about by an impairment of insulin production or its release by the beta cells of the islets of Langerhans. More often it is due to a resistance to the action of insulin either due to a receptor/post receptor defect or an imbalance between insulin and its counter regulatory hormones. Clinically diabetes is characterized by a wide spectrum of disorders ranging from asymptomatic hyperglycemia to abnormalities in the various organs.
PANCREAS
The endocrine component of the pancreas consists of different types of cells: α-cells, β-cells, δ-cells and PP cells contained in the islets of Langerhans which constitute 1% of its weight. There are 100,000 islets in the pancreas, and each islet contains 1000-3000 cells. Thus altogether there are 100-300 million β-cells in the pancreas.
Pancreatic beta cells can store 200 units of insulin and can release 30-50 units of insulin per day. 95% of cells of the pancreas have exocrine function and 5% have endocrine function. The beta cells produce insulin, alpha cells produce glucagon, delta cells produce somatostatin and the PP cells produce pancreatic polypeptide.
CLASSIFICATION
The Classification suggested by American Diabetes Association (ADA) is called as the etiological classification of diabetes and has the two main types of diabetes labeled as type 1 and type 2, along with gestational diabetes mellitus and the other specific types where a precise etiological factor is identified.
The revised diagnostic criteria give equal importance to the fasting and 2 hours post glucose load plasma glucose (2h PG) for the diagnosis of diabetes, thereby eliminating the need for a routine oral glucose tolerance test (OGTT) for the diagnosis of diabetes. The cut-off level of fasting plasma glucose (FPG) for diagnosis of diabetes has been fixed as 126 mg/dl, since this reflects the same degree of hyperglycemia as a 2hr-PG of 200 mg/dl in terms of susceptibility for the development of microvascular and macrovascular complications. These criteria are expected to rationalize and simplify the diagnosis of diabetes and a larger number of people could be screened due to the simplification of the procedure of doing only fasting plasma glucose rather than an OGTT.
ETIOPATHOGENESIS
Type 1Diabetes:
Type-1 diabetes mellitus has been classified into type-1A in which cell-mediated autoimmune attack on the beta cells is more prominent and type-1B in which the mechanism is less clear. Type1B is less frequent of the two.
Classification of type-1 diabetes mellitus
1. Preclinical
a. Autoantibodies + / OGTT normal.
b. Autoantibodies + / OGTT abnormal.
c. Autoantibodies + / fasting hyperglycemia
2. Clinical – with diabetes
1a. Autoantibodies present (autoimmune)
1b. Autoantibodies absent (idiopathic)
3. Explosive onset / fulminent onset
4. Rapid onset
5. Late onset (LADA).
ETIOLOGICAL CLASSIFICATION OF DIABETES MELLITUS
1. Type 1 diabetes (cell destruction, usually leading to absolute insulin deficiency)
• Immune mediated
• Idiopathic
2. Type 2 diabetes (may range from predominantly insulin resistance with relative insulin deficiency to a predominantly secretory defect with insulin resistance)
3. Other specific types
A. Genetic defects of cell function
• Chromosome 12, HNF-1 (MODY 3)
• Chromosome 7, glucokinase (MODY 2)
• Chromosome 20, HNF-4 (MODY 1)
• Mitochondrial DNA
• Others.
B. Genetic defects in insulin action
• Type A insulin resistance
• Leprechaunism
• Rabson-Mendenhall syndrome
• Lipoatrophic diabetes
• Other
C. Diseases of the exocrine pancreas
• Pancreatitis, Trauma, Pancreatectomy
• Neoplasia
• Cystic-Fibrosis, Hemochromatosis
• Fibrocalculous pancreatopathy
• Others
D. Endocrinopathy
• Acromegaly/Cushing’s syndrome
• Glucagonoma, pheochromocytoma
• Hyperthyroidism, somatostatinoma
• Aldosteronoma
• Others
E. Drug or chemical induced
• Pentamidine
• Nicotinic acid
• Glucocorticoids
• Thyroid hormone, diazoxide
• Adrenergic agonists
• Thiazides, phenytoin
• Interferons
• Others. Immunosuppressive drugs, steroids, tarcrolimus, cyclosporin
F. Infections
• Congenital rubella
• Cytomegalovirus
• Others
G. Uncommon forms of immune mediated diabetes
• Stiff man syndrome
• Anti-insulin receptor antibodies
• Others
H. Genetic syndromes associated with diabetes
• Down’s syndrome, Turner’s syndrome
• Klinefelter’s syndrome
• Wolfram’s syndrome, Friedreich’s ataxia
• Huntington’s chorea
• Laurence-Moon-Biedl syndrome
• Myotonic dystrophy, porphyria
• Prader-Willi syndrome
• Others
4. Gestational diabetes mellitus
Type 2 DM
Type 2 DM (previously known as NIDDM) is considered as a ‘multifactorial’ or ‘complex’ disease due to the complex interaction between various genetic and environmental factors in its pathogenesis. Multiple evidence suggests that genetic factors play a major role in this condition. A genetic predisposition running through families is evident. Identical twins invariably develop type 2 diabetes when exposed to the same environmental factors. In genetically predisposed individuals several environmental factors precipitate the onset of diabetes.
Important among these are obesity, physical inactivity, repeated pregnancies, infections, physical or psychological stress and diabetogenic drugs. Birth of large babies weighing above 4 kg is a strong pointer to the subsequent development of diabetes in the mother.
Obesity
The current obesity epidemic due to the modern sedentary life and caloric abundance is a major factor that predisposes to type 2diabetes. Hence it is invariably seen that type 2 diabetes is closely related to obesity. Obese subjects show a relative resistance to the action of insulin due to a reduction in the number of insulin receptors on the target cells. The full complement of receptors is restored on shedding the excess weight.
There is an association between low birth weight in infancy and occurrence of IGT or DM in young adulthood. Increase in the body mass index (BMI) after the age of 2 years is also associated with the chance to develop DM.
Physical Inactivity
It seems to act as a factor favoring the onset of type 2 diabetes. Many of the diabetics are physically inactive. Physical exercise improves their exercise tolerance.
Role of Insulin antagonists: Glucose metabolism is delicately balanced by the coordinated effects of insulin antagonist hormones like glucagon, cortisol, catecholamines and growth hormone. Several other hormones also take part in the metabolism of carbohydrates. Imbalance of this hormonal profile results in carbohydrate intolerance.
Other antagonists to insulin: Antibodies to insulin may develop in individuals who are on treatment with insulins especially the animal insulins. The antibodies inactivate endogenous as well as administered insulin. Such patients show progressive insulin resistance. Fatty acids which compete with carbohydrate for metabolism in muscle lead to insulin resistance. In hyperlipidemia insulin dependent carbohydrate metabolism suffers and a relative insulin resistance develops.
Thus it would seem that persons are predisposed to develop type 2 diabetes by genetically. However lifestyle factors will determine the onset, age of onset, severity of the metabolic defect and further course.
PATHOLOGICAL CHANGES
Pancreas: In type 1 diabetes the beta cells of the islets of Langerhans show reduction in number, degranulation and hyalinization. In recent onset type 1 DM lymphocytic infiltration of the islets occurs and this may be caused by viral infection. Inflammation is seen particularly around the beta cells only and not around the other types of cells.
In type 2 DM during the early phase the beta cells are normal in number or only slightly reduced. The beta cells lose their sensitivity to the hyperglycemic stimulus for releasing insulin. As a result insulin secretion loses its smooth and fine relationship with glucose level. It tends to be erratic. In the early stages of evolution of type 2 DM—the reduction in the sensitivity of the receptors is compensated by overproduction of insulin and accompanying hyperinsulinemia. Frank diabetes results when beta cells starts failing and insulin production comes down.
Insulin resistance in muscle develops early in persons who would develop type 2 diabetes later. Beta cell function starts deteriorating about 10 years before the onset of DM, by which time the beta cell function has fallen to 30% or less. Acanthosis nigricans is a cutaneous marker of hyperinsulinemia. Both impaired fasting glucose (1FG) and
impaired glucose tolerance (IGT) lead to type 2 diabetes in a variable proportion of patients.
Several factors account for the frequency and time of onset of complications in diabetes. These include theabnormalities of glucose levels, genetic factors, smoking, obesity, hypertension, hyperlipidemia and others.
Vascular Changes
Diabetics show a predisposition to develop vascular lesions affecting both small and large blood vessels. In microangiopathy, there is specific involvement of the small blood vessels. Venules, capillaries and arterioles are affected in this process. There is deposition of PAS (periodic acid Schiff) positive material in the capillary basement membrane. Glycosylation of several proteins in the vessel wall results in increased permeability. The basement membrane is thickened. Ultimately there is vascular occlusion.
Microangiopathy is most marked in type 1, developing early in life but also occurs in type 2. Various factors like endothelial damage, increased plasma viscosity, erythrocyte aggregation, reduced red cell deformability and increased platelet adhesion lead to microangiopathy. The problem is more complex and the entire process is still not fully understood. Microangiopathy affects several organ systems. The main lesions are seen in the retina; kidneys, peripheral nerves and heart giving rise to diabetic retinopathy, nephropathy, many forms of diabetic neuropathy and cardiomyopathy.
Macroangiopathy
The diabetic is prone to develop occlusive vascular disease in medium sized arteries such as the coronary, cerebral and peripheral limb vessels. The process is one of atheroma which sets in at younger ages and is more extensive than that occurring in non diabetics. These lesions lead to increased risk of ischemic heart disease, cerebrovascular accidents and ischemia to the limbs with intermittent claudication and peripheral gangrene. Macroangiopathy largely accounts for the steep rise in mortality in middle-aged diabetics.
Retinopathy
Diabetes mellitus produces a classical retinopathy. A specific change occurs in the vessels leading to loss of mural cells (pericytes) and the formation of micro aneurysms. The occurence of retinopathy is related more to the duration of the disease than to the severity. Once initiated the fundus changes are usually progressive. The early changes are venous dilation and the appearance of small dot like micro aneurysms in the perimacular area.
Arterial blood is shunted and this leads to ischemia of the retina. Increased vascular permeability accounts for the formation of exudates. In the next stage dot and blot hemorrhages predominate. Large subhyaloid hemorrhages and vitreous hemorrhages may develop and vision is seriously impaired. Such hemorrhages are due to rupture of newly formed blood vessels. As these hemorrhages are absorbed, organization by fibrous tissue results and multiple bands of retinitis proliferans develop. These lead to permanent visual impairment. The fibrous bands may contract giving rise to retinal detachment. Leaking vessels in the retina can be demonstrated by fluorescein angiography.
Retinopathy is usually associated with advanced nephropathy. Sometimes in diabetic ketoacidosis with severe hyperlipidemia the fat gives a milky white appearance to the retinal arteries called “lipemia retinalis”
Renal Lesions
These are commonly seen in subjects who have had diabetes for over 15-20 years. Vascular changes include (i) arteriosclerosis of the renal artery, (ii) sclerosis of the arterioles and (iii) glomerulosclerosis. Glomerulosclerosis may be nodular (Kimmelstiel-wilson lesion) or diffuse. There is accumulation of PAS positive eosinophilic material within the mesangium. There is thickening of the glomerular capillary basement membrane. The establishment of glomerulosclerosis is indicated by the presence of proteinuria. Further damage to the glomeruli results in the development of chronic renal failure.
Distant stages can be defined in the evolution of diabetic nephropathy. In the initial stages, asymptomatic microalbuminuria in which up to 200 mcg/minute of albumin may be lost in the urine. Normal subjects do not lose more than 20 mcg/min or 300 mg of protein in 24 hours. Microalbuminuria is not detectable by the ordinary laboratory tests. In type 1 DM there is elevation of systotic BP during sleep preceding micro albuminuria. This rise in BP is an important contributory factor in the development of structural changes in the kidneys. It is absolutely necessary to control blood pressure also along with blood sugar to prevent deterioration.
In the early stage the kidneys are enlarged, more vascular and the glomerular filtration rate (GFR) is increased. In the second stage, there is microalbuminuria and in third stage the proteinuria is more pronounced and easily detectable by routine tests. Loss of 3.5g or more of protein in 24 hours may lead to the development of nephrotic syndrome. Hypertension develops during this stage. In the fourth stage further structural changes develop and the glomerular filtration rate comes down with gradual increase in the blood levels of metabolic waste products such as creatinine and urea. The fifth stage is one of gross reduction of glomerular filtration and overt renal failure with azotemia, severe hypertension and complications such as cardiac failure and end stage renal failure. Autonomic neuropathy may lead to functional obstruction of the bladder, retention with over flow, urinary infection and further deterioration of renal functions. Another system of classification is based on creatinine clearance.
The diabetic patient is predisposed to develop urinary infection and therefore acute and chronic pyelonephritis are very common. Fulminant urinary infection leads to ischemic necrosis of the renal papillae. This presents as acute anuric renal failure. Fleshy masses may be passed in the urine. These are the necrosed papillae and the condition is called papillitis necroticans ulcerans. Emphysematouspyelonephritis is another serious complication.
Peripheral Nerves
In the myelinated nerve fibers, axonal atrophy was considered to be the primary lesion, secondary to ineffective axonal transport. Axoglial dysfunction, and abnormalities of paranodal connections between the terminal myelin loops and the axonal membrane have also been described. This could explain the reduction in nerve conduction velocity. This improves with therapeutic inhibition of aldose reductase. More recent studies, however provide evidence for the presence of demyelination and hence Schwann’s cell involvement is the primary lesion. As the myelinated fibers degenerate, there is an attempt to regenerate, which manifests in the form of regeneration clusters. With progress of the neuropathy the density of the regeneration clusters also comes down. Structural abnormalities have also been found in the vessels supplying the nerve fibers. The epineural vessels show arteriolar attenuation, venous distension, arteriovenous shunting and new vessel formation along with intimal hyperplasia and hypertrophy, denervation and reduction in neuropeptide expression. The perineural vessels also demonstrate basement membrane thickening and endothelial cell hypertrophy and hyperplasia. There is also a reduction in capillary density and occurrence of pericyte loss with reduction of endoneural oxygen tension and blood flow to the nerves.
CLINICAL FEATURES
The clinical manifestations of diabetes are protean. Though the symptoms are similar in both types of diabetes, in type 1 they develop acutely whereas in the majority of the type 2 the onset is insidious. Type 1 patients are usually below the age of 30, thin and emaciated and unless promptly treated with insulin, they would develop ketoacidosis.
Due to the high prevalence of obesity, type 2 diabetes is occurring at earlier ages as a global phenomenon, especially in developed countries.
Type 2 patients are generally above the age of 30, obese, usually asymptomatic and may present directly with the vascular complications of diabetes. Around 50% of the cases present with the classical symptoms of polyuria, polyphagia and weight loss. These symptoms can be directly correlated with hyperglycemia and glycosuria. Other clinical presentations which warrant full investigation to exclude diabetes are (i) non-healing ulcers (ii) recurrent respiratory or urinary tract infections (iii) Rapid changes in refraction of the eyes (iv) steady and unexplained rapid weight loss (v) increased tendency for fungal infections like moniliasis, balanoposthitis and vulvitis; (vi) unexplained peripheral neuropathy (vii) premature onset of ischemic heart disease, stroke or vascular occlusions (viii) history of overweight babies and recurrent fetal loss in women (ix) premature cataract often below the age of 50 years and retinopathy (x) impotence in males, and (xi) any vague ill-health. In some cases, diabetics may present to the doctor for the first time with any of the major emergencies, without any apparent illness previously.
DIAGNOSIS
Diabetics with classic symptoms can be diagnosed clinically, but since many cases may be asymptomatic, diabetes should be suspected even in the absence of symptoms. The clinical symptoms and the biochemical alterations do not go hand in hand in many cases. Diabetes being mainly a biochemical disease with several different but inter-related biochemical and molecular abnormalities, should always be diagnosed and managed with biochemical monitoring along with clinical examination.
Fasting plasma glucose levels above 126 mg/dL (6.7 mmol/L) or postprandial plasma glucose levels above 200 mg/dL are diagnostic. It is always better to do bothestimations to confirm the diagnosis.
Estimation of FBS and PPBS has become mandatory investigations in all health check up examinations.
Urine tests: These tests can be used for initial screening and follow-up of cases under treatment. Urinary glucose does not always directly reflect the blood glucose level. The value of urine examination cannot be underestimated since protenuria, ketonuria and the microscopic abnormalities can be detected only by examining the urine.
Glucose in urine is tested by the Benedict’s test and Clinitest (Chemtab), which detect reducing substances non-specifically. While glucose is by far the most common reducing substance in urine, the possibility of other reducing substance should be kept in mind and the enzyme methods (employing glucose oxidase) which are specific for glucose (Clinistix, Diastix) should be employed. If the Benedict’s test is positive and the glucose oxidase is negative, the presence of other reducing substances such as ascorbic acid, aspirin and lactose should be suspected.
Blood glucose estimation: Various methods are employed to estimate the blood glucose. The methods using copper reduction (Folin-Wu or Nelson Somogyi) also detect the reducing substances like uric acid, creatinine and hence their values are 20-30 mg/dL higher than those obtained by the glucose oxidsae method which gives the true glucose values. Highly accurate and rapid (1-2 min) devices are now available based on immobilized glucose oxidase electrodes. Hexokinase and glucose dehydrogenase methods are used for reference. Blood sugar estimations are mandatory for confirming the diagnosis of diabetes. Both fasting and postprandial values should be estimated. In mild diabetes the fasting blood sugar values may be below 126 mg /dL and therefore the diagnosis is likely to be missed if only the fasting blood sugar is estimated.
Glucose tolerance test (GTT): The oral glucose tolerance test (OGTT) is mainly used for diagnosis of diabetes when blood glucose levels are equivocal, during pregnancy, or in an epidemiological setting to screen for diabetes and impaired glucose tolerance.
The OGTT should be administered in the morning after at least 3 days of unrestricted diet (greater than 150 g of carbohydrate daily) and usual physical activity. The test should be preceded by an overnight fast of 8-14 h. during which period only water may be drunk. Smoking is not permitted during the test. The presence of factors such as medications, inactivity and infection that influence interpretation of the results of the test must be recorded.
After collection of the fasting blood sample, the subject should drink 75 g of anhydrous glucose dissolved in 250-300 mLof water over the course of 5 minutes. For children, the test load should be 1.75g of glucose per kg of body weight, up to a maximum of 75g of glucose. Blood samples should be once again collected 2 hr after the test load.
If there is delay in estimation of glucose the blood samples should be collected in a tube containing sodium fluoride (6 mg/mL of whole blood) and immediately centrifuged to separate the plasma. In subjects having symptoms of diabetes, a single fasting value above 126 mg/dL or a 2-hour blood glucose value above 200 mg/dL after 75 g of glucose oral may be taken to be diagnostic. In asymptomatic subjects at least two abnormal blood glucose values should be insisted upon to confirm the clinical diagnosis.
TREATMENT
The aim of treatment is to achieve normal blood glucose levels throughout day and night, to alleviate symptoms and to prevent complications. The four pillars of diabetic management are diet, exercise, drugs and patient education, backed up by regular monitoring of glycemic control and early detection and treatment of complications.
CURRENT ISSUES AND TRENDS IN MHC
CURRENT ISSUES AND TRENDS IN MENTAL HEALTH CARE
UPDATED 2024
A Psychiatric nurse faces various challenges because of changes in the inpatient care approach. Some of these changes that affect her role are as follows:
Trends in Health Care
- Increased mental health problems
- Provision for quality and comprehensive services
- Multidisciplinary team approach
- Providing continuity of care
- Care provided in alternative settings
Economic Issues
- Industrialization
- Urbanization
- Raised standard of living
Changes in Illness Orientation
- Shift from illness to prevention (modification of style), specific to holistic, quantity of care to quality of care
Changes in Care Delivery
- Care delivery is shifted from institutional services to community services, genetic services to counseling services, nurse patient relationship to nurse-patient partnership.
Information Technology
- Telenursing
- Telemedicine
- Mass media
- Electronic systems
- Nursing informatics
Consumer Empowerment
- Increased consumer awareness
- Awareness of the community in early detection and treatment of mental illness as well as proper utilization of available psychiatric hospitals
- Patients are health care consumers demanding quality health care services at affordable cost with less restrictive and more humane rates.
Deinstitutionalization
- Bringing mental health patients out of the hospital and shifting care to community.
Physician Shortage and Gaps in Service
- Physician shortage can provide the opportunity for new roles, for example, nurse practitioner. In respect to gaps in services, nurses always meet the needs of people for whom services are not available, for example, home visiting nurse.
Demographic Changes
- Increasing number of the elderly group
- Type of family (Increased number of nuclear families).
Change in Patient Needs
- Wanting a more holistic orientation in health care.
Challenges in Psychiatric Nursing
- Knowledge development, dissemination and application
- Overcoming stigma
- Health care delivery system issues
- Impact of technology
Educational Programs for the Psychiatric Nurse
- Diploma in Psychiatric Nursing (The first program was offered in 1956 at NIMHANS, Bengaluru)
- MSc in Psychiatric Nursing (The first program was offered in 1976 at Rajkumari Amrit Kaur College of Nursing, New Delhi)
- Mphil in Psychiatric Nursing (1990, MG University, Kottayam)
- Doctorate in Psychiatric Nursing (offered at MAHE, Manipal; RAK College of Nursing, Delhi; NIMHANS, Bengaluru, National Consortium for PhD in Nursing under RGUHS, Karnataka, etc)
- Short term training programs for both the degree and diploma holders in nursing
Standards of Mental Health Nursing
The development of standards for nursing practice is a beginning step towards the attainment of quality nursing care. The adoption of standards helps to clarify nurses areas of accountability, since the standards provide the nurse, the health agency, other professionals, patients, and the public, with a basis for evaluating practice. Standards also define the nursing profession’s accountability to the public. These standards are therefore a means for improving the quality of care for mentally ill people.
Development of Code of Ethics
This is very important for a psychiatric nurse as she takes up independent roles in Psychotherapy, behavior therapy, cognitive therapy, individual therapy, group therapy, maintains patient’s confidentiality, protects his rights and acts as patient’s advocate.
Legal Aspects in Psychiatric Nursing
Knowledge of the legal boundaries governing psychiatric nursing practice is necessary to protect the public, the patient, and the nurse. The practice of psychiatric nursing is influenced by law, particularly in its concern for the rights of patients and the quality of care they receive.
The patient’s right to refuse a particular treatment, protection from confinement, intentional torts, informed consent, confidentiality, and record keeping are a few legal issues in which the nurse has to participate and gain quality knowledge.
Promotion of Research in Mental Health Nursing
The nurse contributes to nursing and the mental health field through innovations in theory and practice and participation in research.
Cost-effective Nursing Care
Studies need to be conducted to find out the viability in terms of cost involved in training a nurse and the quality of output in terms of nursing care rendered by her.
Focus of Care
A psychiatric nurse has to focus care on certain target groups like the elderly, children, women, youth, mentally retarded and chronic mentally ill.

URINE TESTING
Urine Testing Uses – Purpose, Characteristics, Examination, Preliminary Assessment, Equipment, Procedure, Urine pH, Gravity, After Care
UPDATED 2024
Urine testing, also known as urinalysis, is a diagnostic test that involves analyzing a person’s urine for various markers, compounds, and characteristics. This type of testing can provide valuable information about a person’s overall health, help diagnose medical conditions, and monitor the effectiveness of treatments.
USES OF URINE TESTING
Here are some common uses of urine testing:
- Drug Testing: Urine tests are frequently used to screen for the presence of drugs and their metabolites. This is common in workplaces, athletic organizations, and legal situations.
- Medical Conditions: Urinalysis can help diagnose various medical conditions, such as diabetes, kidney diseases, urinary tract infections (UTIs), and liver problems. Abnormal levels of glucose, protein, blood cells, or other substances in the urine may indicate an underlying health issue.
- Pregnancy Testing: Urine tests are often used to detect the presence of human chorionic gonadotropin (hCG), a hormone produced during pregnancy. Home pregnancy tests typically use urine samples for this purpose.
- Kidney Function: Urine tests can provide information about kidney function by measuring levels of creatinine, urea, and other substances. Changes in these levels can indicate kidney problems.
- Metabolic Disorders: Certain metabolic disorders, such as phenylketonuria (PKU) or maple syrup urine disease (MSUD), can be diagnosed through urine testing.
- Monitoring Medications: Some medications can be monitored through urine testing to ensure that they are at therapeutic levels and not causing adverse effects.
URINE TESTING
Urine analysis methods comprise testing reaction, specific gravity, albumen, sugar, bile, acetone, pus, blood and yeasts microscopically
Purpose
- To detect reaction, in cystitis the reaction is alkaline
- To detect sugar, it is present in diabetes mellitus
- To detect – protein it is present in kidney damage, pre-eclampsia and is called proteinuria
- To detect acetone, it is present due to incomplete metabolism of fat
- To detect bile – it is seen in cases of obstructive jaundice or hemolytic diseases
- To detect pus cells – it is present due to urinary tract infection
- To detect blood – it is seen in snake bite, fracture pelvis, etc
Characteristics of Normal Urine
- Volume: 1,000 to 2,000 ml in 24 hours
- Appearance: clear
- Odor: aromatic color
- Color: amber or pale straw in color
- Reaction: normal urine is slightly acidic
- Specific gravity: 1.010 to 1.025
- Constituents of the normal urine: water 96 percent, urea 2% and uric acid, urates, creatinine, chlorides, phosphates, sulfates, oxalates – 2%
Characteristics of Abnormal Urine
Volume
- Polyuria – increased in volume
- Oliguria – decreased in volume
- Anuria – total absence or marked decrease of urine
- Suppression – failure of the kidney to secrete urine
Color
- Green or brownish yellow – bile salts and bile pigments
- Reddish brown – urobilinogen
- Bright red – a large amount of fresh blood
- Smokey brown – blood pigment
- Milk white – chyluria due to filariasis
Appearance
- Mucus – appears as a flocculent cloud
- Pus – settles at the bottom as a heavy cloud
- Stones – as fine sand
- Uric acid – as grains of pepper
Odor
- Sweetish or fruity odor – seen in diabetes
Reaction
- Alkaline – cystitis
- Specific gravity
- Diabetes mellitus – increased specific gravity
- Renal disease – low specific gravity
- Constituents of urine
- Kidney damage – albumin
Types of Examination of the Urine
- Physical examination: color appearance, volume, reaction, specific gravity and color
- Chemical examination: routine tests such as for albumin and sugar. Special tests such as tests for acetone, bile pigments and bile salts. Microscopic examination – crystals, casts, RBC, pus cells, epithelial and bacteria
Preliminary Assessment
- The doctor order for any instructions
- Articles available in the unit
- General condition and diagnosis of the patient
- Self-care ability of the patient
Preparation of the Patient and Environment
- Explain the procedure to the patient
- Keep the urine sample ready
- Arrange the articles ready in the treatment
- Provide labeled container for collecting urine
Equipment
- Test tubes 4 to 6 on a test tube
- Test tube holder – 1
- Spirit lamp – 1
- Match box – 1
- Kidney tray with lining to discard the wastes
- Duster or rag piece – to wipe the outside of the test tube before heating
- Acetic acid – to test urine for albumin
- Nitric acid or sulfosalicyclic acid – to test urine for albumin
- Red and blue litmus paper – to test the reaction of the urine
- Urinometer – to measure the specific gravity of the urine
- Benedict’s solution – to test urine for sugar
- Ammonium sulfate crystals, sodium nitroprusside crystals and liquor ammonia to test urine for acetone
- Weak solution of Tr. Iodine to test for bile pigments
- Sulfur powder: to test for bile salts
- Glass jar: to measure the amount of the urine
- Pipette – 2 – to measure drops of urine and reagents
- A small bottle brush – to clean the test tubes
Procedure
Sugar Test
- Take test tube and fix in holder
- Pour 5 ml of Benedict’s solution into test tube
- Light spirit lamp and heat Benedict solution till it boils
- Holding test tube mouth facing away from nurse
- Add 8 drops of urine using dropper and allow boiling for few seconds
- Put off flame and cool test tube under running water
Observations
- Blue: Nil
- Green: +
- Yellow: ++
- Orange: +++
- Brick red: ++++
Albumin Test
A hot test
- Fill 2/3 of test tube with urine, secure test tube holder at very top
- Heat the upper third of test tube over flame
- If there is precipitation, it denotes the presence of wither protein or phosphate
- Add 2-4 drops of 2 percent acetic acid
- If precipitate dissolves it is due to phosphates present in normal urine
- If precipitate does not dissolve it denotes presence of albumin
Observation
- Trace: +
- Cloudy:++ (100mg/dL)
- Thick cloudiness: +++ (500 g/dL)
Cold Test
- Pour a small quantity of nitric acid or sulfosalicylic acid 3 percent in to a clean test tube
- Allow equal quantity of urine to trickle down the sides of the test tube
- If albumin present, a white precipitate will be seen where two fluids meet
Urine pH
- Collect and keep ready with urine sample
- Dip litmus strip in urine and keep for one minute
- Note color change
- Discard strip into container for infected waste
Urine Specific Gravity
- Fill 3/4 of jar with urine
- Gently place urinometer into jar
- When urinometer stops bobbing
- Read specific gravity directly from scale marked on calibrated stem of urinometer
- Make sure that instrument floats freely and does not touch sides of jar
- Read scale at lowest point of meniscus to ensure an accurate reading at eye level
Rothera’s Test (Acetone)
- Take 2 cm depth of ammonium sulfate crystals in a small test tube
- Add equal volume of urine and one crystal of sodium nitroprusside
- Close the test tube with a cork and shake the test tube
- Take liquor ammonia and add it to the urine, trickling through the sides
- Read the results immediately
Observations
If acetone is present permanganate purple colored ring is formed at the junction of urine and ammonia
Hays Test (Bile Salts)
- Take a test tube, half full of urine
- Sprinkle sulfur powder on the surface of the urine
- If the powder sinks down to the test tube, it indicates the presence of bile salts
Smith’s Test (Bile Pigments)
- Fill 3/4 of test tube with urine
- Add iodine drops along the sides of the tube, so as to form a layer on the surface of the urine
- A green color at the junction of the two liquids indicates the presence of bile pigments
After Care
- Discard the urine in the sluice room
- Wash the test tube with soap and water
- Dry the tube, holder and urinometer with jar
- Replace the article after cleaning
- Wash hands thoroughly
Record the procedure in the nurse’s record sheet and dietetic chart

TEMPERATURE TECHNIQUES
TEMPERATURE TECHNIQUES – Principles, Equipment and Procedure (COMMUNITY HEALTH NURSING)
UPDATED 2024
A clinical thermometer is a special instrument designed to measure the temperature of the body. Two thermometers – one oral and one rectal – are essential equipment which the nurse always carries in her bag. Elevation in temperature is an indication that the body is reacting to an infection
PRINCIPLES
- Meticulous cleaning of thermometer before and after use is essential to prevent the spread of infection
- Temperature is usually taken by mouth. Rectal temperatures are most accurate while auxillary temperatures are least accurate
- Shake the mercury to 95 degree F before taking the temperature
- Keep all thermometers in the shade and in the coolest part of the building
- Accuracy in temperature helps in effective treatment and medical decision
EQUIPMENT
- Ten small cotton swabs
- Kidney basin to hold moist cotton swabs
- Thermometer
- Lubricant for rectal temperature
- Paper bag
PROCEDURE
- Use bag technique as per standard precautions
- Remove the thermometer swab up to bulb and read
- Place the thermometer at proper site
- Rinse the thermometer thoroughly under cool running water after reading
- Replace it in the community bag and wash hands
- Record it in the TPR sheet

CHOLECYSTOGRAPHY
CHOLECYSTOGRAPHY – Purpose, Preparation of the Patient, Procedure, After Care and Complications
UPDATED 2024
Cholecystography is a test for gallbladder disease, done by visualizing the gallbladder. Visualization of the gallbladder depends upon absorption of the dye from the intestinal tract, isolation and excretion by the liver cells and a free passage way from the liver to the gallbladder.
PURPOSE
- To detect gallstones
- To test the contractibility of the gallbladder
- To find out filling ability of the gallbladder
- To find out its ability to concentrate, its contents, and its condition when it is empty in normal states
PREPARATION OF THE PATIENT
- Explain the procedure to the patient to relieve tension and worries
- Stop medications which contain iodine compounds and bismuth three days prior to the test
- Check whether the patient is allergic to iodine or sea food before giving the dye
- Record the patient’s weight to calculate the dose of the dye
- The patient is given a low-fat evening meal to avoid gallbladder contraction. Thereafter, no food and water should be given to the patient until the X-ray examinations are complete
- The bowel is cleansed with saline enema
- The emergency drugs and resuscitation equipment should be kept ready to resuscitate the patient
PROCEDURE
- The patient is given a light diet at 7 pm without fat
- A laxative like dulcolax is given to clear the bowels
- At 10 pm the patient is given 6 telepaque tablets to swallow
- This dye is opaque to X-rays and is absorbed from the intestines, and is excreted by the liver
- When the gallbladder is normal, this dye gets concentrated in the gallbladder, which becomes visible by X-ray
- X-ray pictures are taken on the following day approximately 14, 18 and 19 hours after the drug has been administered, i.e. at 12 noon. 4 pm and 5 pm
- No food is given during this period
- Then, to test the contractibility of the gallbladder, the patient is fed with a fatty meal, one hour before the last X-rays taken at 12 noon and 4 pm, but it empties itself after the fatty meal taken at 4 pm and is, therefore, not visible in the X-ray taken at 5 pm
- An abnormal gallbladder may not get filled properly or may fail to empty itself
AFTER CARE
- Observe the patient for allergic reactions. Check the vital signs of the patient
- Accompany the patient throughout the procedure
- Make the patient comfortable
COMPLICATIONS
Severe reactions to dye may lead to:
- Respiratory difficulty
- Urticaria
- Shock
- Collapse

THERAPEUTIC COMMUNICATION
THERAPEUTIC COMMUNICATION – Principles or Characteristics, Techniques and Non-therapeutic Communication
UPDATED 2024
Therapeutic communication is an interpersonal interaction between the nurse and the patient during which the nurse focuses on the patient’s specific needs to promote an effective exchange of information. All nurses need skills in therapeutic communication to effectively apply the nursing process and to meet standards of care for their patients.
Therapeutic communication can help nurses to accomplish many goals:
- Establish a therapeutic nurse-patient relationship
- Identify the most important patient’s needs
- Assess the patient’s perception of the problem
- Facilitate the patient’s expression of emotions
- Implement interventions designed to address the patient’s needs
To have an effective therapeutic communication, the nurse must consider privacy and respect of boundaries, use of touch and active listening and observation.
Principles or Characteristics of Therapeutic Communication
- The patient should be the primary focus of interaction
- A professional attitude sets the tone of the therapeutic relationship
- Use self-disclosure cautiously and only when it has a therapeutic purpose
- Avoid social relationship with patients
- Maintain patient confidentiality
- Assess the patient’s intellectual competence to determine the level of understanding
- Implement interventions from a theoretic base
- Maintain a nonjudgmental attitude. Avoid making judgments about patient’s behavior
- Avoid giving advice
- Guide the patient to reinterpret his or her experiences rationally.
Therapeutic Communication Techniques
- Listening: It is an active process of receiving information. Responses on the part of the nurse, such as maintaining eye-to-eye contact, nodding, gesturing and other forms of receptive nonverbal communication convey to the patient that he is being listened to and understood.
Therapeutic Value: Nonverbally communicates to the patient the nurse’s interest and acceptance.
- Broad Openings: Encouraging the patient to select topics for discussion. For example, “What are you thinking about?”
Therapeutic Value: Indicates acceptance by the nurse and the value of patient’s initiative.
- Restating: Repeating the main thought expressed by the patient. For example, “You say that your mother left you when you were 5-year-old”
Therapeutic Value: Indicated that the nurse is listening and validates, reinforces or calls attention to something important that has been said.
- Clarification: Attempting to put vague ideas or unclear thoughts of the patient into words to enhance the nurse’s understanding or asking the patient to explain what he means. For example, “I am not sure what you mean. Could you tell me about the again?”
Therapeutic Value: It helps to clarify feelings, ideas and perceptions of the patient and provides an explicit correlation between them and the patient’s actions.
- Reflection: Directing back the patient’s ideas, feelings, questions and content. For example, “You are feeling tense and anxious and it is related to a conversation you had with your husband last night”.
Therapeutic Value: Validates the nurse’s understanding of what the patient is saying and signifies empathy, interest and respect for the patient.
- Humor: The discharge of energy through comic enjoyment of the imperfect. For example, “That gives a whole new meaning to the word ‘nervous’”, said with shared kidding between the nurse and the patient.
Therapeutic Value: Can promote insight by making repressed material conscious, resolving paradoxes, tempering aggression and revealing new options, and is a socially acceptable for of sublimation.
- Informing: The skill of information giving. For example,” I think you need to know more about your medications.”
Therapeutic Value: Helpful in health teaching or patient education about relevant aspects of patient’s well-being and self-care.
- Focusing: Questions or statements that help the patient expand on a topic of importance. For example, “I think that we should talk more about your relationship with your father”.
Therapeutic Value: Allows the patient to discuss central issues and keeps the communication process goal-directed.
- Sharing Perceptions: Asking the patient to verify the nurses understanding of what the patient is thinking or feeling. For example, ”You are smiling, but I sense that you are really very angry with me”.
Therapeutic Value: Conveys the nurse understands to the patient and has the potential for clearing up confusing communication.
- Theme Identification: This involves identification of underlying issues or problems experienced by the patient that emerge repeatedly during the course of the nurse-patient relationship. For example, “I noticed that you said, you have been hurt or rejected by the man. Do you think this is an underlying issue?”
Therapeutic Value: It allows the nurse to promote the patient’s exploration and understanding of important problems.
- Silence: Lack of verbal communication for a therapeutic reason. For example, sitting with a patient and nonverbally communicating interest and involvement.
Therapeutic Value: Allows the patient time to think and gain insight, slows the pace of the interaction and encourages the patient to initiate conversation while enjoying the nurse’s support, understanding and acceptance.
- Suggesting: Presentation of alternative ideas for the patient’s consideration relative to problem solving. For example, “Have you thought about responding to your boss in a different way when he raises that issue with you? You could ask him if a specific problem has occurred.”
Therapeutic Value: Increases the patient’s perceived notions or choices.
Ineffective/Non-therapeutic Communication
These include failure to listen, conflicting verbal or non-verbal messages, a judgmental attitude, false reassurance, giving of advice, the inability to receive information because of a preoccupation of impaired thought process and changing of the subject if one becomes uncomfortable with the topic being discussed.


