DIABETES MELLITUS – General Characteristics, Pancreas, Classification, Etiopathogenesis, Pathological Changes, Clinical Features, Diagnosis and Treatment
UPDATED 2024
General Characteristics
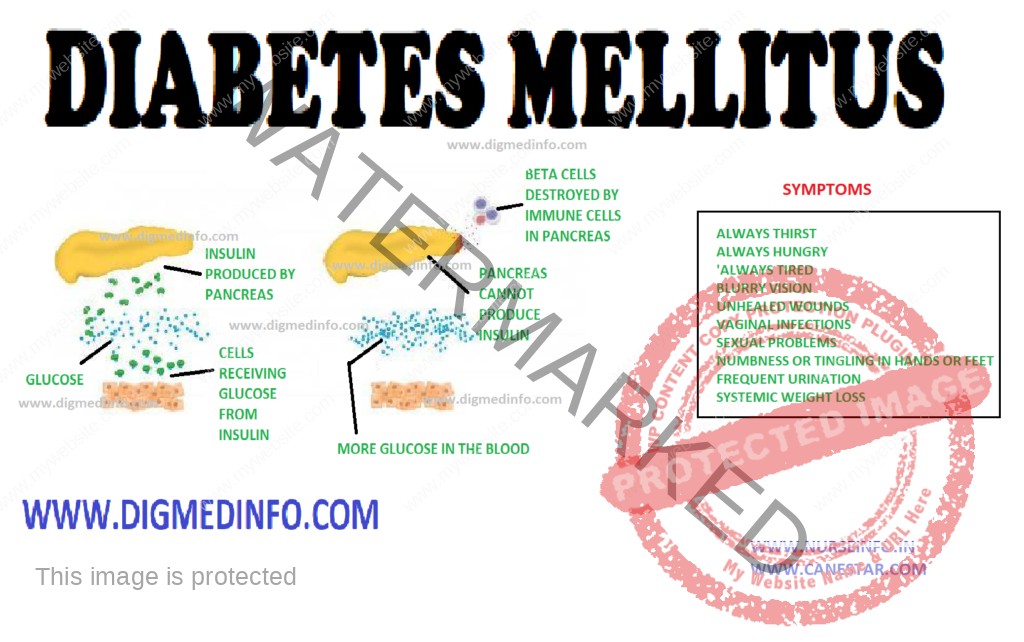
Diabetes mellitus is a chronic metabolic disorder characterized by hyperglycemia with or without glycosuria, resulting from an absolute or relative deficiency of insulin. This is brought about by an impairment of insulin production or its release by the beta cells of the islets of Langerhans. More often it is due to a resistance to the action of insulin either due to a receptor/post receptor defect or an imbalance between insulin and its counter regulatory hormones. Clinically diabetes is characterized by a wide spectrum of disorders ranging from asymptomatic hyperglycemia to abnormalities in the various organs.
PANCREAS
The endocrine component of the pancreas consists of different types of cells: α-cells, β-cells, δ-cells and PP cells contained in the islets of Langerhans which constitute 1% of its weight. There are 100,000 islets in the pancreas, and each islet contains 1000-3000 cells. Thus altogether there are 100-300 million β-cells in the pancreas.
Pancreatic beta cells can store 200 units of insulin and can release 30-50 units of insulin per day. 95% of cells of the pancreas have exocrine function and 5% have endocrine function. The beta cells produce insulin, alpha cells produce glucagon, delta cells produce somatostatin and the PP cells produce pancreatic polypeptide.
CLASSIFICATION
The Classification suggested by American Diabetes Association (ADA) is called as the etiological classification of diabetes and has the two main types of diabetes labeled as type 1 and type 2, along with gestational diabetes mellitus and the other specific types where a precise etiological factor is identified.
The revised diagnostic criteria give equal importance to the fasting and 2 hours post glucose load plasma glucose (2h PG) for the diagnosis of diabetes, thereby eliminating the need for a routine oral glucose tolerance test (OGTT) for the diagnosis of diabetes. The cut-off level of fasting plasma glucose (FPG) for diagnosis of diabetes has been fixed as 126 mg/dl, since this reflects the same degree of hyperglycemia as a 2hr-PG of 200 mg/dl in terms of susceptibility for the development of microvascular and macrovascular complications. These criteria are expected to rationalize and simplify the diagnosis of diabetes and a larger number of people could be screened due to the simplification of the procedure of doing only fasting plasma glucose rather than an OGTT.
ETIOPATHOGENESIS
Type 1Diabetes:
Type-1 diabetes mellitus has been classified into type-1A in which cell-mediated autoimmune attack on the beta cells is more prominent and type-1B in which the mechanism is less clear. Type1B is less frequent of the two.
Classification of type-1 diabetes mellitus
1. Preclinical
a. Autoantibodies + / OGTT normal.
b. Autoantibodies + / OGTT abnormal.
c. Autoantibodies + / fasting hyperglycemia
2. Clinical – with diabetes
1a. Autoantibodies present (autoimmune)
1b. Autoantibodies absent (idiopathic)
3. Explosive onset / fulminent onset
4. Rapid onset
5. Late onset (LADA).
ETIOLOGICAL CLASSIFICATION OF DIABETES MELLITUS
1. Type 1 diabetes (cell destruction, usually leading to absolute insulin deficiency)
• Immune mediated
• Idiopathic
2. Type 2 diabetes (may range from predominantly insulin resistance with relative insulin deficiency to a predominantly secretory defect with insulin resistance)
3. Other specific types
A. Genetic defects of cell function
• Chromosome 12, HNF-1 (MODY 3)
• Chromosome 7, glucokinase (MODY 2)
• Chromosome 20, HNF-4 (MODY 1)
• Mitochondrial DNA
• Others.
B. Genetic defects in insulin action
• Type A insulin resistance
• Leprechaunism
• Rabson-Mendenhall syndrome
• Lipoatrophic diabetes
• Other
C. Diseases of the exocrine pancreas
• Pancreatitis, Trauma, Pancreatectomy
• Neoplasia
• Cystic-Fibrosis, Hemochromatosis
• Fibrocalculous pancreatopathy
• Others
D. Endocrinopathy
• Acromegaly/Cushing’s syndrome
• Glucagonoma, pheochromocytoma
• Hyperthyroidism, somatostatinoma
• Aldosteronoma
• Others
E. Drug or chemical induced
• Pentamidine
• Nicotinic acid
• Glucocorticoids
• Thyroid hormone, diazoxide
• Adrenergic agonists
• Thiazides, phenytoin
• Interferons
• Others. Immunosuppressive drugs, steroids, tarcrolimus, cyclosporin
F. Infections
• Congenital rubella
• Cytomegalovirus
• Others
G. Uncommon forms of immune mediated diabetes
• Stiff man syndrome
• Anti-insulin receptor antibodies
• Others
H. Genetic syndromes associated with diabetes
• Down’s syndrome, Turner’s syndrome
• Klinefelter’s syndrome
• Wolfram’s syndrome, Friedreich’s ataxia
• Huntington’s chorea
• Laurence-Moon-Biedl syndrome
• Myotonic dystrophy, porphyria
• Prader-Willi syndrome
• Others
4. Gestational diabetes mellitus
Type 2 DM
Type 2 DM (previously known as NIDDM) is considered as a ‘multifactorial’ or ‘complex’ disease due to the complex interaction between various genetic and environmental factors in its pathogenesis. Multiple evidence suggests that genetic factors play a major role in this condition. A genetic predisposition running through families is evident. Identical twins invariably develop type 2 diabetes when exposed to the same environmental factors. In genetically predisposed individuals several environmental factors precipitate the onset of diabetes.
Important among these are obesity, physical inactivity, repeated pregnancies, infections, physical or psychological stress and diabetogenic drugs. Birth of large babies weighing above 4 kg is a strong pointer to the subsequent development of diabetes in the mother.
Obesity
The current obesity epidemic due to the modern sedentary life and caloric abundance is a major factor that predisposes to type 2diabetes. Hence it is invariably seen that type 2 diabetes is closely related to obesity. Obese subjects show a relative resistance to the action of insulin due to a reduction in the number of insulin receptors on the target cells. The full complement of receptors is restored on shedding the excess weight.
There is an association between low birth weight in infancy and occurrence of IGT or DM in young adulthood. Increase in the body mass index (BMI) after the age of 2 years is also associated with the chance to develop DM.
Physical Inactivity
It seems to act as a factor favoring the onset of type 2 diabetes. Many of the diabetics are physically inactive. Physical exercise improves their exercise tolerance.
Role of Insulin antagonists: Glucose metabolism is delicately balanced by the coordinated effects of insulin antagonist hormones like glucagon, cortisol, catecholamines and growth hormone. Several other hormones also take part in the metabolism of carbohydrates. Imbalance of this hormonal profile results in carbohydrate intolerance.
Other antagonists to insulin: Antibodies to insulin may develop in individuals who are on treatment with insulins especially the animal insulins. The antibodies inactivate endogenous as well as administered insulin. Such patients show progressive insulin resistance. Fatty acids which compete with carbohydrate for metabolism in muscle lead to insulin resistance. In hyperlipidemia insulin dependent carbohydrate metabolism suffers and a relative insulin resistance develops.
Thus it would seem that persons are predisposed to develop type 2 diabetes by genetically. However lifestyle factors will determine the onset, age of onset, severity of the metabolic defect and further course.
PATHOLOGICAL CHANGES
Pancreas: In type 1 diabetes the beta cells of the islets of Langerhans show reduction in number, degranulation and hyalinization. In recent onset type 1 DM lymphocytic infiltration of the islets occurs and this may be caused by viral infection. Inflammation is seen particularly around the beta cells only and not around the other types of cells.
In type 2 DM during the early phase the beta cells are normal in number or only slightly reduced. The beta cells lose their sensitivity to the hyperglycemic stimulus for releasing insulin. As a result insulin secretion loses its smooth and fine relationship with glucose level. It tends to be erratic. In the early stages of evolution of type 2 DM—the reduction in the sensitivity of the receptors is compensated by overproduction of insulin and accompanying hyperinsulinemia. Frank diabetes results when beta cells starts failing and insulin production comes down.
Insulin resistance in muscle develops early in persons who would develop type 2 diabetes later. Beta cell function starts deteriorating about 10 years before the onset of DM, by which time the beta cell function has fallen to 30% or less. Acanthosis nigricans is a cutaneous marker of hyperinsulinemia. Both impaired fasting glucose (1FG) and
impaired glucose tolerance (IGT) lead to type 2 diabetes in a variable proportion of patients.
Several factors account for the frequency and time of onset of complications in diabetes. These include theabnormalities of glucose levels, genetic factors, smoking, obesity, hypertension, hyperlipidemia and others.
Vascular Changes
Diabetics show a predisposition to develop vascular lesions affecting both small and large blood vessels. In microangiopathy, there is specific involvement of the small blood vessels. Venules, capillaries and arterioles are affected in this process. There is deposition of PAS (periodic acid Schiff) positive material in the capillary basement membrane. Glycosylation of several proteins in the vessel wall results in increased permeability. The basement membrane is thickened. Ultimately there is vascular occlusion.
Microangiopathy is most marked in type 1, developing early in life but also occurs in type 2. Various factors like endothelial damage, increased plasma viscosity, erythrocyte aggregation, reduced red cell deformability and increased platelet adhesion lead to microangiopathy. The problem is more complex and the entire process is still not fully understood. Microangiopathy affects several organ systems. The main lesions are seen in the retina; kidneys, peripheral nerves and heart giving rise to diabetic retinopathy, nephropathy, many forms of diabetic neuropathy and cardiomyopathy.
Macroangiopathy
The diabetic is prone to develop occlusive vascular disease in medium sized arteries such as the coronary, cerebral and peripheral limb vessels. The process is one of atheroma which sets in at younger ages and is more extensive than that occurring in non diabetics. These lesions lead to increased risk of ischemic heart disease, cerebrovascular accidents and ischemia to the limbs with intermittent claudication and peripheral gangrene. Macroangiopathy largely accounts for the steep rise in mortality in middle-aged diabetics.
Retinopathy
Diabetes mellitus produces a classical retinopathy. A specific change occurs in the vessels leading to loss of mural cells (pericytes) and the formation of micro aneurysms. The occurence of retinopathy is related more to the duration of the disease than to the severity. Once initiated the fundus changes are usually progressive. The early changes are venous dilation and the appearance of small dot like micro aneurysms in the perimacular area.
Arterial blood is shunted and this leads to ischemia of the retina. Increased vascular permeability accounts for the formation of exudates. In the next stage dot and blot hemorrhages predominate. Large subhyaloid hemorrhages and vitreous hemorrhages may develop and vision is seriously impaired. Such hemorrhages are due to rupture of newly formed blood vessels. As these hemorrhages are absorbed, organization by fibrous tissue results and multiple bands of retinitis proliferans develop. These lead to permanent visual impairment. The fibrous bands may contract giving rise to retinal detachment. Leaking vessels in the retina can be demonstrated by fluorescein angiography.
Retinopathy is usually associated with advanced nephropathy. Sometimes in diabetic ketoacidosis with severe hyperlipidemia the fat gives a milky white appearance to the retinal arteries called “lipemia retinalis”
Renal Lesions
These are commonly seen in subjects who have had diabetes for over 15-20 years. Vascular changes include (i) arteriosclerosis of the renal artery, (ii) sclerosis of the arterioles and (iii) glomerulosclerosis. Glomerulosclerosis may be nodular (Kimmelstiel-wilson lesion) or diffuse. There is accumulation of PAS positive eosinophilic material within the mesangium. There is thickening of the glomerular capillary basement membrane. The establishment of glomerulosclerosis is indicated by the presence of proteinuria. Further damage to the glomeruli results in the development of chronic renal failure.
Distant stages can be defined in the evolution of diabetic nephropathy. In the initial stages, asymptomatic microalbuminuria in which up to 200 mcg/minute of albumin may be lost in the urine. Normal subjects do not lose more than 20 mcg/min or 300 mg of protein in 24 hours. Microalbuminuria is not detectable by the ordinary laboratory tests. In type 1 DM there is elevation of systotic BP during sleep preceding micro albuminuria. This rise in BP is an important contributory factor in the development of structural changes in the kidneys. It is absolutely necessary to control blood pressure also along with blood sugar to prevent deterioration.
In the early stage the kidneys are enlarged, more vascular and the glomerular filtration rate (GFR) is increased. In the second stage, there is microalbuminuria and in third stage the proteinuria is more pronounced and easily detectable by routine tests. Loss of 3.5g or more of protein in 24 hours may lead to the development of nephrotic syndrome. Hypertension develops during this stage. In the fourth stage further structural changes develop and the glomerular filtration rate comes down with gradual increase in the blood levels of metabolic waste products such as creatinine and urea. The fifth stage is one of gross reduction of glomerular filtration and overt renal failure with azotemia, severe hypertension and complications such as cardiac failure and end stage renal failure. Autonomic neuropathy may lead to functional obstruction of the bladder, retention with over flow, urinary infection and further deterioration of renal functions. Another system of classification is based on creatinine clearance.
The diabetic patient is predisposed to develop urinary infection and therefore acute and chronic pyelonephritis are very common. Fulminant urinary infection leads to ischemic necrosis of the renal papillae. This presents as acute anuric renal failure. Fleshy masses may be passed in the urine. These are the necrosed papillae and the condition is called papillitis necroticans ulcerans. Emphysematouspyelonephritis is another serious complication.
Peripheral Nerves
In the myelinated nerve fibers, axonal atrophy was considered to be the primary lesion, secondary to ineffective axonal transport. Axoglial dysfunction, and abnormalities of paranodal connections between the terminal myelin loops and the axonal membrane have also been described. This could explain the reduction in nerve conduction velocity. This improves with therapeutic inhibition of aldose reductase. More recent studies, however provide evidence for the presence of demyelination and hence Schwann’s cell involvement is the primary lesion. As the myelinated fibers degenerate, there is an attempt to regenerate, which manifests in the form of regeneration clusters. With progress of the neuropathy the density of the regeneration clusters also comes down. Structural abnormalities have also been found in the vessels supplying the nerve fibers. The epineural vessels show arteriolar attenuation, venous distension, arteriovenous shunting and new vessel formation along with intimal hyperplasia and hypertrophy, denervation and reduction in neuropeptide expression. The perineural vessels also demonstrate basement membrane thickening and endothelial cell hypertrophy and hyperplasia. There is also a reduction in capillary density and occurrence of pericyte loss with reduction of endoneural oxygen tension and blood flow to the nerves.
CLINICAL FEATURES
The clinical manifestations of diabetes are protean. Though the symptoms are similar in both types of diabetes, in type 1 they develop acutely whereas in the majority of the type 2 the onset is insidious. Type 1 patients are usually below the age of 30, thin and emaciated and unless promptly treated with insulin, they would develop ketoacidosis.
Due to the high prevalence of obesity, type 2 diabetes is occurring at earlier ages as a global phenomenon, especially in developed countries.
Type 2 patients are generally above the age of 30, obese, usually asymptomatic and may present directly with the vascular complications of diabetes. Around 50% of the cases present with the classical symptoms of polyuria, polyphagia and weight loss. These symptoms can be directly correlated with hyperglycemia and glycosuria. Other clinical presentations which warrant full investigation to exclude diabetes are (i) non-healing ulcers (ii) recurrent respiratory or urinary tract infections (iii) Rapid changes in refraction of the eyes (iv) steady and unexplained rapid weight loss (v) increased tendency for fungal infections like moniliasis, balanoposthitis and vulvitis; (vi) unexplained peripheral neuropathy (vii) premature onset of ischemic heart disease, stroke or vascular occlusions (viii) history of overweight babies and recurrent fetal loss in women (ix) premature cataract often below the age of 50 years and retinopathy (x) impotence in males, and (xi) any vague ill-health. In some cases, diabetics may present to the doctor for the first time with any of the major emergencies, without any apparent illness previously.
DIAGNOSIS
Diabetics with classic symptoms can be diagnosed clinically, but since many cases may be asymptomatic, diabetes should be suspected even in the absence of symptoms. The clinical symptoms and the biochemical alterations do not go hand in hand in many cases. Diabetes being mainly a biochemical disease with several different but inter-related biochemical and molecular abnormalities, should always be diagnosed and managed with biochemical monitoring along with clinical examination.
Fasting plasma glucose levels above 126 mg/dL (6.7 mmol/L) or postprandial plasma glucose levels above 200 mg/dL are diagnostic. It is always better to do bothestimations to confirm the diagnosis.
Estimation of FBS and PPBS has become mandatory investigations in all health check up examinations.
Urine tests: These tests can be used for initial screening and follow-up of cases under treatment. Urinary glucose does not always directly reflect the blood glucose level. The value of urine examination cannot be underestimated since protenuria, ketonuria and the microscopic abnormalities can be detected only by examining the urine.
Glucose in urine is tested by the Benedict’s test and Clinitest (Chemtab), which detect reducing substances non-specifically. While glucose is by far the most common reducing substance in urine, the possibility of other reducing substance should be kept in mind and the enzyme methods (employing glucose oxidase) which are specific for glucose (Clinistix, Diastix) should be employed. If the Benedict’s test is positive and the glucose oxidase is negative, the presence of other reducing substances such as ascorbic acid, aspirin and lactose should be suspected.
Blood glucose estimation: Various methods are employed to estimate the blood glucose. The methods using copper reduction (Folin-Wu or Nelson Somogyi) also detect the reducing substances like uric acid, creatinine and hence their values are 20-30 mg/dL higher than those obtained by the glucose oxidsae method which gives the true glucose values. Highly accurate and rapid (1-2 min) devices are now available based on immobilized glucose oxidase electrodes. Hexokinase and glucose dehydrogenase methods are used for reference. Blood sugar estimations are mandatory for confirming the diagnosis of diabetes. Both fasting and postprandial values should be estimated. In mild diabetes the fasting blood sugar values may be below 126 mg /dL and therefore the diagnosis is likely to be missed if only the fasting blood sugar is estimated.
Glucose tolerance test (GTT): The oral glucose tolerance test (OGTT) is mainly used for diagnosis of diabetes when blood glucose levels are equivocal, during pregnancy, or in an epidemiological setting to screen for diabetes and impaired glucose tolerance.
The OGTT should be administered in the morning after at least 3 days of unrestricted diet (greater than 150 g of carbohydrate daily) and usual physical activity. The test should be preceded by an overnight fast of 8-14 h. during which period only water may be drunk. Smoking is not permitted during the test. The presence of factors such as medications, inactivity and infection that influence interpretation of the results of the test must be recorded.
After collection of the fasting blood sample, the subject should drink 75 g of anhydrous glucose dissolved in 250-300 mLof water over the course of 5 minutes. For children, the test load should be 1.75g of glucose per kg of body weight, up to a maximum of 75g of glucose. Blood samples should be once again collected 2 hr after the test load.
If there is delay in estimation of glucose the blood samples should be collected in a tube containing sodium fluoride (6 mg/mL of whole blood) and immediately centrifuged to separate the plasma. In subjects having symptoms of diabetes, a single fasting value above 126 mg/dL or a 2-hour blood glucose value above 200 mg/dL after 75 g of glucose oral may be taken to be diagnostic. In asymptomatic subjects at least two abnormal blood glucose values should be insisted upon to confirm the clinical diagnosis.
TREATMENT
The aim of treatment is to achieve normal blood glucose levels throughout day and night, to alleviate symptoms and to prevent complications. The four pillars of diabetic management are diet, exercise, drugs and patient education, backed up by regular monitoring of glycemic control and early detection and treatment of complications.