PROTEIN ENERGY MALNUTRITION – General Characteristics, Etiology and Pathogenesis, Clinical Features (Kwashiorkor and Marasmus)
GENERAL CHARACTERISTICS
Protein-energy malnutrition (PEM) has been defined by WHO and FAO (1973) as “a range of pathological conditions arises from coincident lack of protein and calories in varying proportions occurring most frequently in infants and young children and commonly associated with infections”.
The clinical spectrum includes a variety of clinical syndromes, with ‘marasmus’ and ‘kwashiorkor’ at the two extremes and the intermediate forms (marasmic kwashiorkor) and nutritional dwarfism in between. Mild cases may present only with apathy and retardation of growth. Childhood malnutrition has been graded taking the weight for age as the criteria.
Etiology and pathogenesis:
Protein-energy malnutrition is an environmental disease, caused by deficient intake of protein and calories. It results from the combined influences of low food availability, poverty, ignorance, illiteracy and cultural taboos, frequent infections and poor environmental sanitation.
CLINICAL FEATURES
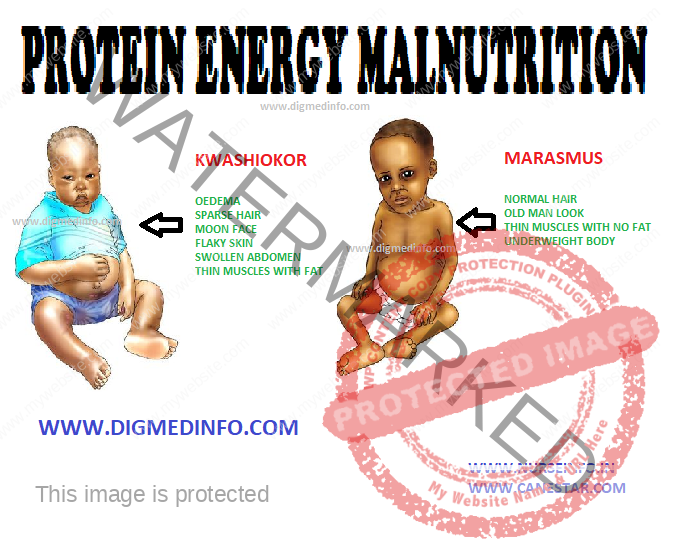
Marasmus
Severe restriction of food in an infant, as in cases of gross inadequacy of breast milk, leads to arrest of growth. Due to starvation the subcutaneous fat and muscles are used up as energy sources. The baby becomes emaciated. Unlike as in kwashiorkor, there is no edema. Loss of the buccal pads of fat give the infant a withered and ‘old man’s’ look. The weight is below the sixtieth percentile. The height depends upon the onset and duration of undernutrition. In the early stages the infant eats well but appetite is lost as the condition progresses. Initially, inadequacy of food leads to constipation, but later on diarrhea sets in with green stools containing mucus. In the well-established form the baby is apathetic.
Moderate anemia may develop. The skin and hair are usually normal.
KWASHIORKOR
This name was coined by Dr Cicely Williams in 1933, to denote ‘disease of the child deposed from the breast by the conception of a new fetus’. Children between the ages of 1 and 3 years are affected more. The disease starts when the baby is weaned from the breast.
Pathology
Main changes are seen in the small intestines, liver, pancreas and thymus. Small intestinal mucosa shows blunting of villi and atrophy of brush border, so that the columnar epithelium appears to become cuboidal. The total absorbing surface is reduced. Lactose intolerance is common because of disaccharidase deficiency. Liver shows fatty infiltration of the parenchymal cells. The pancreatic acini are atrophic and enzyme activity is reduced. Thymus is markedly atrophied and this may contribute to deficiency of cell mediated immunity.
Clinical features
This child is stunted and skeletal muscles are wasted. The presence of fairly normal amounts of subcutaneous fat and edema give a deceptively plump appearance. Pitting edema is a prominent feature.
The child is apathetic, irritable and drowsy. Characteristic skin changes occur in many and when present; these are diagnostic. These include flaky paint dermatosis seen over areas of pressure and trauma, fissuring and ulceration at the flexures and a mosaic like appearance (crazy pavement appearance). The hair becomes thin, sparse, brownish and lusterless. They may fall off. Regrowth of normal pigmented hair heralds nutritional recovery. Since periods of nutritional deprivation and partial correction of nutrition alternate in many cases, the hair shows alternate bands of pigmentation and depigmentation (flag sign). The appetite is poor. Diarrhea is a frequent feature. There may be concomitant deficiencies of iron, folate, fat soluble vitamins and B complex factors. Hepatomegaly occurs in a third of the cases. This is due to fatty infiltration. With recovery the liver reverts to normal without sequelae.
MARASMIC KWASHIORKOR
In this condition, features of marasmus and kwashiorkor are present simultaneously. The body weight is less than 60% of the normal. Dependent edema is present. Mental changes, skin and hair changes and hepatomegaly are evident.
Secondary infection is very common in protein energy malnutrition. This is due to the fact that both humoral and cellular immunity are defective. The intestinal flora is altered and this may account for the diarrhea. Episodes of infection further jeopardize the nutritional status.
Laboratory investigations
The total protein content of the body is reduced, and this is reflected most prominently as hypoalbuminemia. The plasma levels of essential aminoacids are low, but the non-essential amino acids remain normal or even elevated. Basal metabolic rate is reduced. Hypoglycemia occurs commonly. Total body water is increased and all the compartments show increase of fluid. The plasma osmolality is reduced. Renal plasma flow is diminished and this results in impairment of renal function. Plasma sodium is increased with reduction in potassium and magnesium. Gross reduction of serum sodium is associated with a poor prognosis.
Prognosis depends on the severity of the disease at diagnosis and promptness of treatment. Marked weight loss, severe infections, fluid and electrolyte imbalance, hypoglycemia, hypothermia, cardiac failure, elevation of serum bilirubin and liver enzymes, drowsiness and xerophthalmia indicate poor prognosis. In severe cases mortality goes up to 20%. The disease is entirely preventable by ensuring adequate caloric and protein intake to the growing child in the first three years of life. Proper management in the established case gives rise to full recovery, catch up growth and restoration of normal parameters. Recurrence is likely if the adverse environment is not corrected.
Treatment
Treatment is aimed to supply a diet rich in calories, proteins and other essential nutrients. For success of treatment supplementation should be with natural foods available locally. 150 Kcal/kg of energy and 3.3 g/kg of proteins are optimally required for catch-up growth. Fats are administered to supply adequate calories without increasing the bulk. Children with lactose intolerance do not tolerate carbohydrates, but they tolerate fats. As the child gains appetite the frequency of feeding is increased. If there is severe anorexia, forced feeding has to be
resorted to. Salt should be restricted to avoid congestive cardiac failure. The National Institute of Nutrition, Hyderabad has formulated an energy-protein rich mixture to treat PEM at home. It consists of whole wheat 40 g, Bengal gram 16 g, groundnut 10 g, Jaggery 20 g (total 86 g). This supplies 330 calories and 11.3 g proteins. Skimmed milk, eggs and cereals are added as the condition improves. In addition to nutritional correction, intercurrent infections have to be controlled simultaneously. Initial recovery is heralded by the disappearance of edema. The major biochemical abnormalities are corrected within 2-3 weeks. Complete correction of all reversible changes, referred to as ‘clinical recovery’ occurs only in 2-3 months.
Prevention and Rehabilitation
PEM is a preventable disease. Three levels of prevention have been formulated. Primary prevention is achieved by nutrition education to prevent occurrence of PEM. Secondary prevention is aimed at early detection and proper treatment. Tertiary prevention consists of nutritional rehabilitation of an established case. The importance of breast feeding as a prophylactic against protein calorie malnutrition cannot be overemphasized. Correct feeding practices, both during health and disease, proper sanitation, deworming and family planning methods have to be employed simultaneously to achieve lasting benefit. Nutrition rehabilitation is employed to prevent residual nutritional handicap and prevent recurrence of protein energy malnutrition. Even at the initial stages of treatment the mother should be made to participate in the selection, preparation and administration of food. This measure helps to impart nutritional education to her, prevent recurrence and detect relapse early.