CHRONIC KIDNEY DISEASE – DEFINITION AND STAGING, ETIOLOGY, NATURAL HISTORY AND PROGRESSION, CLINICAL MANIFESTATIONS, DIAGNOSIS AND MANAGEMENT
GENERAL CHARACTERISTICS
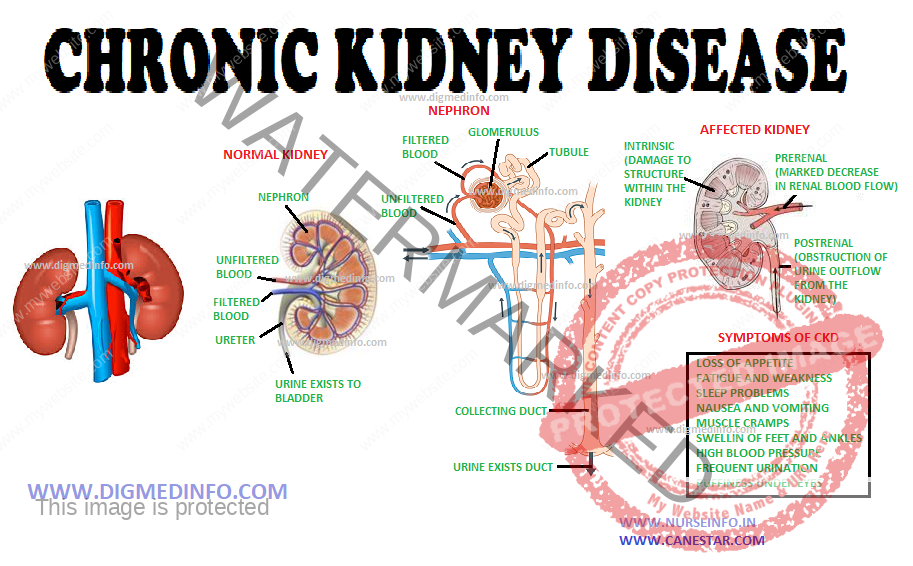
Chronic kidney disease (CKD) is an important public health problem affecting more than 10% of the general population. CKD is an important risk factor progressing to end stage renal disease (ESRD), cardiovascular disease and premature mortality. Management of ESRD is expensive and beyond the reach of many sections of the population in developing countries. CKD is a silent disease and if not detected and treated early, may progress to ESRD. Hence, it is important to identify CKD early and institute measures to retard its progression.
DEFINITION AND STAGING
Chronic kidney disease is defined as either kidney damage or GFR <60mL/min/1.73m2 for >3 months. Kidney damage is defined as pathologic abnormalities or markers of damage, including abnormalities in blood or urine tests or imaging studies.
CKD is characterized by kidney damage characterized by structural, functional or pathological abnormalities in the kidney associated with abnormalities in the composition of urine and/or blood of more than 3 months duration. Anomalies in the imaging tests and decrease in the GFR may be demonstrable. If the GFR is less than 60 mL/min/1.73 m2 body surface, persisting for more than 3 months, this by itself can constitute CKD.
STAGES OF CHRONIC KIDNEY DISEASE
Stage Description GFR (mL/min/1.73m2)
1. Kidney damage with normal > 90
or increased GFR
2. Kidney damage with mild 60-89
decrease in GFR
3. Moderate decrease in GFR 30-59
4. Severe decrease in GFR 15-29
5. Kidney failure < 15 or dialysis
Staging
Stage 1 CKD represents kidney damage when GFR is normal or high. This includes patients with albuminuria or those with abnormal imaging studies.
In stage 2 CKD, there is evidence of kidney damage with mild decrease in GFR (60- 89 mL/min/1.73 m2). In the absence of kidney damage, this level of GFR per se, does not constitute CKD.
Stage 3 CKD includes patients with moderate decline in GFR (59-30 mL/ min/ 1.73 m2). All patients with GFR of less than 60 mL/min/1.73 m2 are classified as having CKD irrespective of whether kidney damage is present or not. This is the stage where serum creatinine starts to rise. Majority of patients still remain asymptomatic. Nocturia and polyuria are early symptoms that may appear at this stage.
In stage 4 CKD, the fall in GFR is severe (29-15 mL/min/1.73 m2). Overt uremic symptoms like loss of appetite, nausea, anemia and recurrent infections occur during this stage.
Stage 5 CKD is the stage when GFR is less than 15 mL/min/ 1.73 m2. At this stage the patient usually requires dialysis or other forms of renal replacement therapy.
GFR is the best measure of overall kidney function in health and disease. GFR can be affected by chronic kidney disease which reduces the number of nephrons. Hemodynamic factors affect single nephron GFR and they result in fall in the GFR. The normal GFR of a young adult is around 120-130 mL/min/1.73 m2. Normal values in women are 8% lower from those of men at all ages. Above the age of 30 years, GFR declines at the rate of approximately 1 mL/min/1.73 m2 per year.
ETIOLOGY
The causes of CKD are broadly classified as given below.
Glomerular Diseases
Primary—Focal segmental glomerulosclerosis (FSGS), IgA nephropathy, membranoproliferative glomerulonephritis (MPGN)
Secondary—Diabetes, systemic lupus erythematosus (SLE)
Tubulointerstitial diseases—Reflux nephropathy, obstructive uropathy.
Vascular diseases—Hypertensive nephrosclerosis, ischemic nephropathy
Cystic diseases—Autosomal dominant and recessive polycystic kidney disease (ADPKD and ARPKD)
Heredofamilial—Alport’s syndrome
Diseases in the transplant -Chronic allograft nephropathy
Age, ethnicity and distribution of renal diseases may be different in various geographical regions. Diabetes and hypertension are the two most important causes of CKD worldwide. In the developing countries chronic glomerulonephritis and chronic interstitial nephritis are relatively more common than in the developed countries.
FSGS, IgA nephropathy, reflux nephropathy and congenital anomalies of the urinary tract lead to CRF usually before the age of 40 years. Diabetes, hypertensive nephrosclerosis, ischemic nephropathy, multiple myeloma, analgesic nephropathy and systemic vasculitis are more common above the age of 55 years.
NATURAL HISTORY AND PROGRESSION OF CKD
Many diseases can intiate kidney injury and lead to progression. Reduction in the number of functioning nephrons lead to hypertrophy and increased glomerular filtration by the surviving nephrons. Glomerular hyperfiltration initially maintains GFR but later, the overworked nephrons succumb to the increased workload and lead to proteinuria, which causes further damage to the glomeruli and the tubulo-interstium. The extent of tubulo-interstitial damage is an important factor determining disease progression. Hypertension which is present in over 90% of cases of advanced CKD leads to further progression of the disease, if not controlled. Hyperglycemia also causes structural and functional changes in the glomeruli leading to progressive renal failure. Progressive destruction of nephrons leads to several adaptive mechanisms which enable the remaining nephrons to maintain body homeostasis. Though these adaptive changes are beneficial, later, they lead to maladaptive consequences. For example, early in the course of renal failure, loss of nephrons result in reduction of phosphate excretion and lead to hyperphosphatemia.
This causes a fall in serum calcium due to its binding to the excess phosphorus. Hypocalcemia stimulates PTH which results in reduction of tubular reabsorption of phosphate and increased phosphorus excretion. This helps to normalize serum phosphate levels. With progressive loss of nephrons, higher and more sustained levels PTH develop leading to secondary hyperparathyroidism with all its adverse consequences. The nature of the primary kidney disease and its activity also dictates the progression. The rate of progression of glomerular diseases is faster than that of tubulo-interstitial diseases.
CLINICAL MANIFESTATIONS OF CKD
Renal impairment is associated with a variety of signs and symptoms that are collectively referred to as the uremic state. The symptomatology may involve any system of the body. However, there is no correlation between the development of symptoms and the severity of renal disease. The early stages of CKD are usually asymptomatic and are detected only on investigations.
Edema
Most of the glomerular diseases are associated with edema. In chronic glomerular disorders, development of intermittent edema and hematuria indicates disease activity. In disorders that are not primarily due to glomerular disorders, as in most tubulointerstitial diseases, edema appears late.
Hypertension
Hypertension is the most common manifestation and it may appear early during the course of renal disease (90% in glomerular and 30% in tubulointerstitial diseases). If untreated, it leads to further damage. Rarely, uncontrolled hypertension can lead to precipitous and irreversible reduction in GFR. Hypertension is also an independent risk factor for cardiovascular morbidity.
Cardiovascular Manifestations
Cardiovascular manifestations include left ventricular hypertrophy (LVH) due to hypertension and anemia, ischemic heart disease, congestive cardiac failure due to fluid overload and myocardial dysfunction due to uremia (uremic cardiomyopathy). Electrolyte disturbances, particularly hyperkalemia can lead to bradycardia, syncope and cardiac arrythmias. Other manifestations include premature atherosclerosis, vascular calcification due to secondary hyperparathyroidism and pericarditis. Pericarditis presents with chest pain. Loud pericardial friction rub is audible on auscultation. Sometimes, fibrinous pericarditis may lead to pericardial effusion and cardiac tamponade. CKD is a major risk factor for cardiovascular disease and even mild impairment of renal function can lead to cardiovascular morbidity and mortality. Conversely, compromise in cardiac function worsens renal function too.
Gastrointestinal Manifestations
The GI manifestations like anorexia, nausea, vomiting, dyspeptic symptoms, constipation or diarrhea usually appear in Stage 4 CKD (GFR between 15 and 30 mL/min). The symptoms are mainly due to gastrointestinal mucosal ulcerations. Uremic stomatitis with dry mucous membranes, multiple small oral ulcers and parotitis are seen in advanced uremia. Rarely, gastrointestinal hemorrhage may occur. ‘Uremic fetor’ describes the ammoniacal odor occurring in patients with advanced renal failure and it is due to the hydrolysis of urea in saliva by bacterial urease. Intractable hiccoughs may occur in advanced uremia. Severe abdominal pain and paralytic ileus may occur as a result of hypokalemia. Ascites may occur in advanced renal failure.
Neuropsychiatric Manifestations
Paresthesias, sensory or motor peripheral neuropathy, pruritus, restless legs syndrome and bladder dysfunction are the neurologic manifestations. Subtle to gross behavioral abnormalities such as anxiety, depression, personality changes and disturbances of sleep are the psychiatric manifestations.
Flapping tremor (asterixis) and myoclonic jerks are features of uncontrolled uremia. Rarely, convulsions and coma may occur. When a patient is initiated on dialysis, abrupt and rapid removal of urea from the blood may lead to higher concentration of urea in the brain since it does not cross the blood-brain barrier rapidly. This leads to cerebral edema and transient neuropsychiatric manifestations, collectively called ‘dialysis disequillibrium syndrome’. This can be prevented by adjusting the initial few sessions of dialysis so as to achieve very gradual fall in blood urea. Patients on long term dialysis may develop features of dementia
and this is attributed to aluminum intoxication which may occur due to intake of aluminum containing antacids or through the water used for dialysis. This is no longer a major problem at present because of water treatment which involves removal of contaminants like aluminium in dialysis water and withdrawal of use of aluminum hydroxide as a phosphate binder.
Cutaneous Manifestations
The characteristic sallow complexion in renal failure is due to pallor and the deposition of yellowish brown urochrome pigment. Recurrent skin infections, dry scaly skin with severe itching, rashes, erythema, vesicles and ulcerations are common. Cutaneous calcification in association with secondary hyperparathyroidism contributes to the severe itching. Pruritic hyperkeratotic papular eruptions or Kyrle’s disease occurs in diabetics. In stage V CKD, precipitation of urea on the surface of the skin gives rise to ‘uremic frost’. Bleeding into the skin and mucosa may occur as a result of platelet dysfunction. Nail changes include pitting, burrowing and ‘half and half’ nails. In half and half nail, the distal half of the nail is pink or brown and the proximal half is white or pale. The conjunctival deposition of calcium leads to redness and gritty feeling in the eye also called the ‘uremic red eye’ while deposition of calcium as a band in the lamina propria of the cornea leads to ‘band keratopathy.’
Hematological Manifestations
Anemia is common in chronic kidney disease. It appears when the GFR is below 50 mL/min (Stage 3 CKD) and progressively worsens as GFR declines further. Symptoms appear much later, (stage 4 CKD). Usually the anemia is normocytic normochromic. The most important cause is decreased secretion of erythropoietin (EPO). Other factors, apart from EPO deficiency which contribute to renal anemia include the following.
1. Circulating uremic toxins – bone marrow resistance to the effect of EPO.
2. Reduced RBC survival (120 days to 80 days) probably due to mild hemolysis.
3. Platelet dysfunction – Bleeding, including occult gastrointestinal blood loss.
4. Iron deficiency.
5. Hyperparathyroidism – bone marrow suppression or fibrosis.
6. Folic acid deficiency.
7. Chronic inflammation.
8. Aluminum toxicity (rare).
Prolonged anemia can lead to worsening of renal function and lead to left ventricular hypertrophy and increased myocardial oxygen demand. Disturbances in the coagulation system and platelet dysfunction are common in advanced uremia. Platelet dysfunction occurs due to factors like retention of uremic toxins, nitric oxide and hyperparathyroidism.
Skeletal Abnormalities – Renal Osteodystrophy
As renal failure progresses, hyperphosphatemia, hypokalemia and secondary hyperparathyroidism develop leading to skeletal abnormalities. Renal tubular defects and altered vitamin D metabolism also contributes to skeletal changes of renal osteodystrophy. Stunting of growth, bone deformities and rickets occur due to end organ resistance to hormones as a result of circulating uremic toxins. Adults with advanced CKD manifest with high turnover bone disease (due to hyperparathyroidism), low turnover bone disease (due to vitamin D deficiency or aluminum toxicity) or adynamic bone disease (due to excessive suppression of PTH). Skeletal deformities are more pronounced especially in children. These manifest even before there is significant reduction of GFR. This is called non-uremic renal osteodystrophy. This is caused by renal tubular acidosis and disturbances in Vitamin D metabolism. With advancing renal failure, bone disease and growth retardation become more evident. Short stature is an important feature of chronic kidney disease of childhood. Uremic toxins and end-organ resistance to growth hormone/insulin like growth factor have been implicated. Prolonged use of corticosteroids may also contribute to growth retardation.
Children with advanced chronic renal failure caused by chronic tubulointerstitial and glomerular diseases also develop severe rickets with deformities (Uremic renal osteodystrophy). In adults, the most common skeletal disturbance is hyperparathyroid bone disease (osteitis fibrosa) characterized by increased osteoclastic bone resorption. This is known as high turnover bone disease. Typical radiographic features include subperiosteal resorption in the phalanges, ‘salt and pepper” pattern in the skull, vascular calcifications in peripheral arteries like radial and femoral arteries and osteosclerosis (rugger jersey spine). Large osteoclastic tumours may be seen (brown tumours) in the skeleton around weight bearing areas. A rare and unusual syndrome in patients with severe osteitis is calciphylaxis. This is due to extra-osseous calcium deposition in soft tissues. It manifests as painful violaceous mottling of the skin followed by progressive gangrenous ulcerations at the fingers, toes and ankles. Osteomalacia is the second pattern of bone disease seen in chronic kidney disease. Vitamin D deficiency and/or aluminum intoxication occurring in those receiving long-term hemodialysis aggravate this condition. It is characterized by severe bone pain, recurring fractures and proximal myopathy. In some cases, a mixed pattern of hyperparathyroidism and osteomalacia may be found. In addition, adynamic bone disease is being increasingly recognized. It is a histological diagnosis showing lack of bone formation and resorption. It is probably due to excessive suppression of PTH by calcium supplements and Vitamin D therapy.
Respiratory Manifestations
Patients with advanced kidney disease may develop dyspnea due to pulmonary edema, pleural effusion or severe metabolic acidosis. Flash pulmonary edema occurs in patients with renovascular diseases due to accelerated hypertension. ‘Uremic lung’ may be seen radiologically as a butterfly shadow in the area of the hilum and this is due to noncardiogenic pulmonary edema associated with increased pulmonary capillary permeability and exudation of proteinaceous fluid into the alveoli. Uremic serositis may present as pleurisy with associated pleural friction rub or underlying hemorrhagic pleural effusion.
DIAGNOSIS
Often CKD may go unnoticed until renal failure is advanced. Strong clinical suspicion and appropriate investigations are essential for early diagnosis. The diagnosis is established by demonstrating decreased GFR or markers of kidney damage. Estimation of serum creatinine and a creatinine clearance are usually used for this purpose.
Markers of kidney damage include proteinuria, hematuria or abnormalities of urinary sediment. Since elevation of serum creatinine occurs only when GFR is below 50% of normal, early detection of CKD depends on the other markers.
Detection of more than 5 erythrocytes per high power field in a freshly voided specimen of urine indicates significant microscopic hematuria if present on repeated examinations. Presence of dysmorphic RBCs and acanthocytes in urine usually indicates pathology in the glomerulus. Coexistence of proteinuria and cellular casts points to renal parenchymal disease. Urinary specific gravity and osmolality may be relatively fixed at around 1010 and 290 respectively, signifying the inability of the kidneys to concentrate or dilute urine. Low serum calcium, high serum phosphorus with high serum alkaline phosphatase is often seen in advanced kidney disease. Serum bicarbonate is often reduced as a result of metabolic acidosis. High anion gap is due to decreased ammonium ion production and decreased excretion of hydrogen ions. Serum levels of sodium are usually normal until renal failure is very advanced. The ability of the kidney to maintain serum potassium homeostasis is preserved till the patient is in stage IV CKD. Hyperkalemia may set in early in patients consuming excessive potassium in diet and those taking potassium sparing diuretics, ACE inhibitors or angiotensin receptor blockers (ARBs).
X-ray of the skeleton may show features of renal rickets in children or osteomalacia, osteitis fibrosa or osteosclerosis in adults. Presence of smaller kidneys detected by ultrasound imaging suggests long standing kidney disease. The exceptions to this rule include diabetes mellitus, multiple myeloma, polycystic kidney disease and obstructive uropathy. Demonstration of scarring of the kidneys may be the earliest indicator of parenchymal damage in diseases like reflux nephropathy. Asymmetry of the kidneys on ultrasound examination may be an indicator of underlying renovascular disease. Calcification and stone disease may be demonstrated by plain X-ray KUB or by ultrasound. Doppler imaging of the kidneys and renal blood vessels helps to detect renovascular disease early.
MANAGEMENT OF CHRONIC KIDNEY DISEASE
All patients require conservative management in the early stages. When the patient approaches Stage V CKD, renal replacement therapy has to be introduced.
The general management consists of:
1. Treatment of reversible causes.
2. Preventing or slowing the progression of the renal disease.
3. Treatment of the complications of renal dysfunction.
4. Identification and adequate preparation for renal replacement therapy.